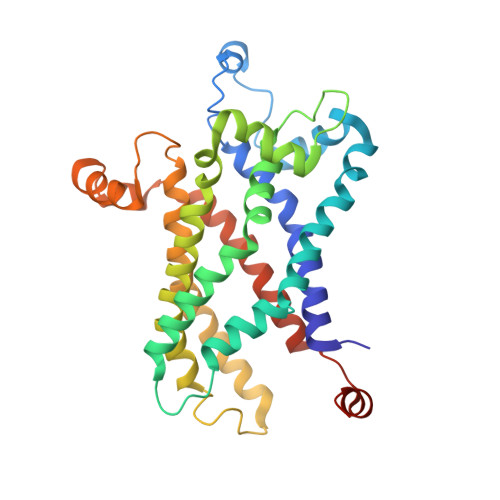
Mitochondrial uncoupling protein 2 structure determined by NMR molecular fragment searching.
Berardi, M.J., Shih, W.M., Harrison, S.C., Chou, J.J.(2011) Nature 476: 109-113
- PubMed: 21785437 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nature10257
- Primary Citation Related Structures:
2LCK - PubMed Abstract:
Mitochondrial uncoupling protein 2 (UCP2) is an integral membrane protein in the mitochondrial anion carrier protein family, the members of which facilitate the transport of small molecules across the mitochondrial inner membrane. When the mitochondrial respiratory complex pumps protons from the mitochondrial matrix to the intermembrane space, it builds up an electrochemical potential. A fraction of this electrochemical potential is dissipated as heat, in a process involving leakage of protons back to the matrix. This leakage, or 'uncoupling' of the proton electrochemical potential, is mediated primarily by uncoupling proteins. However, the mechanism of UCP-mediated proton translocation across the lipid bilayer is unknown. Here we describe a solution-NMR method for structural characterization of UCP2. The method, which overcomes some of the challenges associated with membrane-protein structure determination, combines orientation restraints derived from NMR residual dipolar couplings (RDCs) and semiquantitative distance restraints from paramagnetic relaxation enhancement (PRE) measurements. The local and secondary structures of the protein were determined by piecing together molecular fragments from the Protein Data Bank that best fit experimental RDCs from samples weakly aligned in a DNA nanotube liquid crystal. The RDCs also determine the relative orientation of the secondary structural segments, and the PRE restraints provide their spatial arrangement in the tertiary fold. UCP2 closely resembles the bovine ADP/ATP carrier (the only carrier protein of known structure), but the relative orientations of the helical segments are different, resulting in a wider opening on the matrix side of the inner membrane. Moreover, the nitroxide-labelled GDP binds inside the channel and seems to be closer to transmembrane helices 1-4. We believe that this biophysical approach can be applied to other membrane proteins and, in particular, to other mitochondrial carriers, not only for structure determination but also to characterize various conformational states of these proteins linked to substrate transport.
- Jack and Eileen Connors Structural Biology Laboratory, Harvard Medical School, Boston, Massachusetts 02115, USA.
Organizational Affiliation: