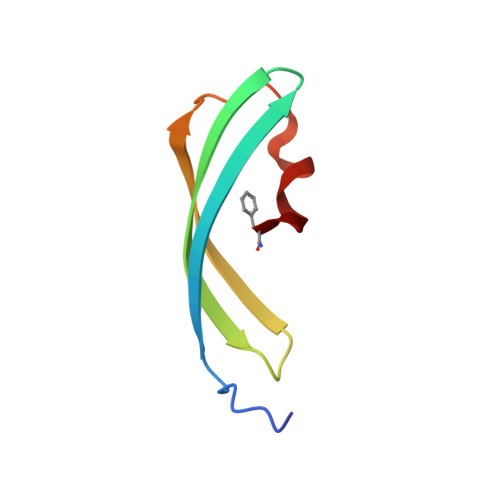
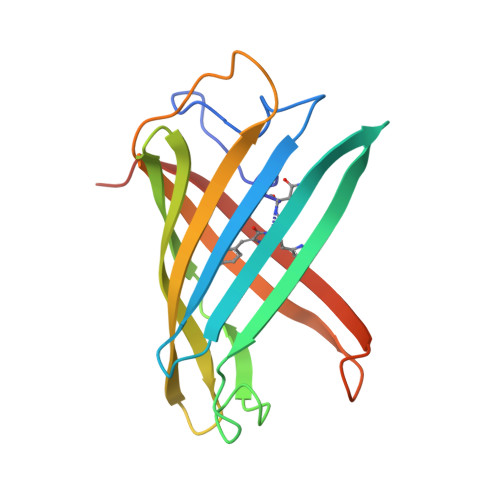
Crystallographic evidence for water-assisted photo-induced peptide cleavage in the stony coral fluorescent protein Kaede.
Hayashi, I., Mizuno, H., Tong, K.I., Furuta, T., Tanaka, F., Yoshimura, M., Miyawaki, A., Ikura, M.(2007) J Mol Biology 372: 918-926
- PubMed: 17692334 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2007.06.037
- Primary Citation Related Structures:
2GW3, 2GW4 - PubMed Abstract:
A coral fluorescent protein from Trachyphyllia geoffroyi, Kaede, possesses a tripeptide of His62-Tyr63-Gly64, which forms a chromophore with green fluorescence. This chromophore's fluorescence turns red following UV light irradiation. We have previously shown that such photoconversion is achieved by a formal beta-elimination reaction, which results in a cleavage of the peptide bond found between the amide nitrogen and the alpha-carbon at His62. However, the stereochemical arrangement of the chromophore and the precise structural basis for this reaction mechanism previously remained unknown. Here, we report the crystal structures of the green and red form of Kaede at 1.4 A and 1.6 A resolutions, respectively. Our structures depict the cleaved peptide bond in the red form. The chromophore conformations both in the green and red forms are similar, except a well-defined water molecule in the proximity of the His62 imidazole ring in the green form. We propose a molecular mechanism for green-to-red photoconversion, which is assisted by the water molecule.
- Division of Signaling Biology, Ontario Cancer Institute and Department of Medical Biophysics, 101 College St., Toronto, Ontario, M5G 1L7, Canada.
Organizational Affiliation: