Identification of a Novel Nonstructural Protein, VP9, from White Spot Syndrome Virus: Its Structure Reveals a Ferredoxin Fold with Specific Metal Binding Sites
Liu, Y., Wu, J.L., Song, J.X., Sivaraman, J., Hew, C.L.(2006) J Virol 80: 10419-10427
- PubMed: 16956937
- DOI: https://doi.org/10.1128/JVI.00698-06
- Primary Citation of Related Structures:
2GJ2, 2GJI - PubMed Abstract:
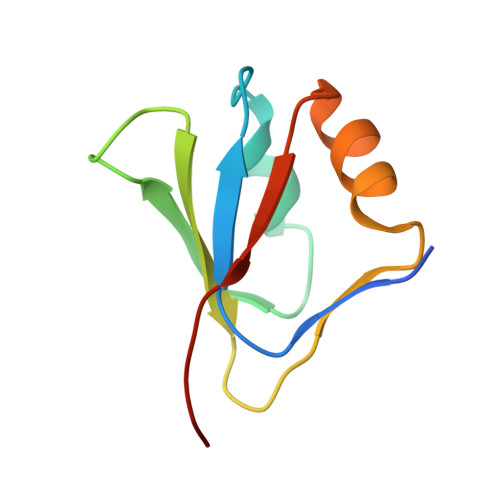
White spot syndrome virus (WSSV) is a major pathogen in shrimp aquaculture. VP9, a full-length protein of WSSV, encoded by open reading frame wsv230, was identified for the first time in the infected Penaeus monodon shrimp tissues, gill, and stomach as a novel, nonstructural protein by Western blotting, mass spectrometry, and immunoelectron microscopy. Real-time reverse transcription-PCR demonstrated that the transcription of VP9 started from the early to the late stage of WSSV infection as a major mRNA species. The structure of full-length VP9 was determined by both X-ray and nuclear magnetic resonance (NMR) techniques. It is the first structure to be reported for WSSV proteins. The crystal structure of VP9 revealed a ferredoxin fold with divalent metal ion binding sites. Cadmium sulfate was found to be essential for crystallization. The Cd2+ ions were bound between the monomer interfaces of the homodimer. Various divalent metal ions have been titrated against VP9, and their interactions were analyzed using NMR spectroscopy. The titration data indicated that VP9 binds with both Zn2+ and Cd2+. VP9 adopts a similar fold as the DNA binding domain of the papillomavirus E2 protein. Based on our present investigations, we hypothesize that VP9 might be involved in the transcriptional regulation of WSSV, a function similar to that of the E2 protein during papillomavirus infection of the host cells.
- Department of Biological Sciences, National University of Singapore, 14 Science Drive 4, Singapore, 117543.
Organizational Affiliation: