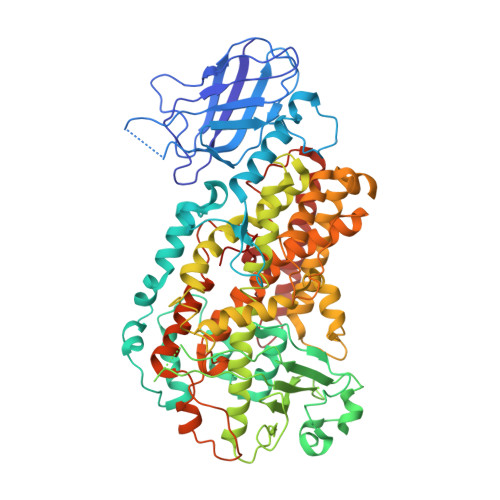
Insights from the X-ray crystal structure of coral 8R-lipoxygenase: calcium activation via a C2-like domain and a structural basis of product chirality.
Oldham, M.L., Brash, A.R., Newcomer, M.E.(2005) J Biological Chem 280: 39545-39552
- PubMed: 16162493
- DOI: https://doi.org/10.1074/jbc.M506675200
- Primary Citation Related Structures:
2FNQ - PubMed Abstract:
Lipoxygenases (LOXs) catalyze the regio- and stereospecific dioxygenation of polyunsaturated membrane-embedded fatty acids. We report here the 3.2 A resolution structure of 8R-LOX from the Caribbean sea whip coral Plexaura homomalla, a LOX isozyme with calcium dependence and the uncommon R chiral stereospecificity. Structural and spectroscopic analyses demonstrated calcium binding in a C2-like membrane-binding domain, illuminating the function of similar amino acids in calcium-activated mammalian 5-LOX, the key enzyme in the pathway to the pro-inflammatory leukotrienes. Mutation of Ca(2+)-ligating amino acids in 8R-LOX resulted not only in a diminished capacity to bind membranes, as monitored by fluorescence resonance energy transfer, but also in an associated loss of Ca(2+)-regulated enzyme activity. Moreover, a structural basis for R chiral specificity is also revealed; creation of a small oxygen pocket next to Gly(428) (Ala in all S-LOX isozymes) promoted C-8 oxygenation with R chirality on the activated fatty acid substrate.
- Department of Biological Sciences, Louisiana State University, Baton Rouge, Louisiana 70803, USA.
Organizational Affiliation: