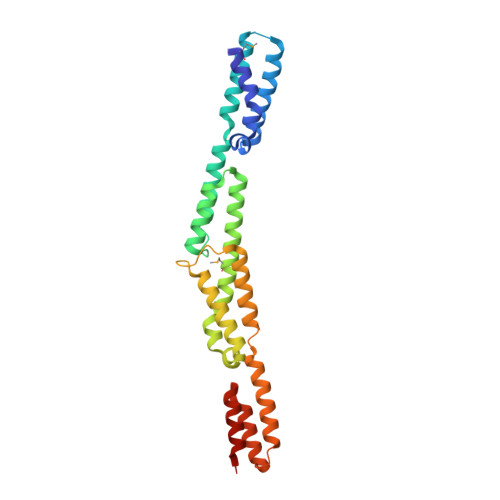
A helical string of alternately connected three-helix bundles for the cell wall-associated adhesion protein Ebh from Staphylococcus aureus
Tanaka, Y., Sakamoto, S., Kuroda, M., Goda, S., Gao, Y.-G., Tsumoto, K., Hiragi, Y., Yao, M., Watanabe, N., Ohta, T., Tanaka, I.(2008) Structure 16: 488-496
- PubMed: 18334223 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2007.12.018
- Primary Citation Related Structures:
2DGJ - PubMed Abstract:
The 1.1 MDa cell-wall-associated adhesion protein of staphylococci, Ebh, consists of several distinct regions, including a large central region with 52 imperfect repeats of 126 amino acid residues. We investigated the structure of this giant molecule by X-ray crystallography, circular dichroism (CD) spectrometry, and small-angle X-ray scattering (SAXS). The crystal structure of two repeats showed that each repeat consists of two distinct three-helix bundles, and two such repeats are connected along the long axis, resulting in a rod-like structure that is 120 A in length. CD and SAXS analyses of the samples with longer repeats suggested that each repeat has an identical structure, and that such repeats are connected tandemly to form a rod-like structure in solution, the length of which increased proportionately with the number of repeating units. On the basis of these results, it was proposed that Ebh is a 320 nm rod-like molecule with some plasticity at module junctions.
- Faculty of Advanced Life Sciences, Hokkaido University, Sapporo 060-0810, Japan.
Organizational Affiliation: