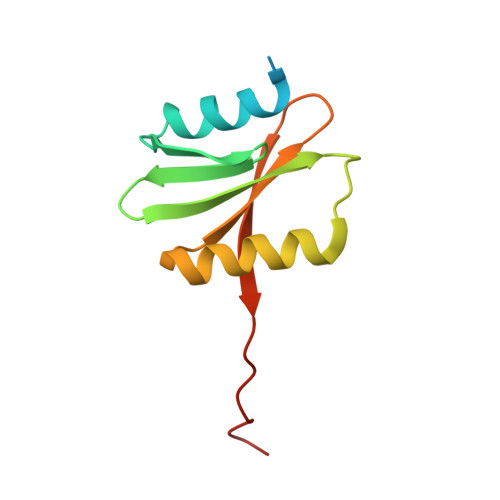
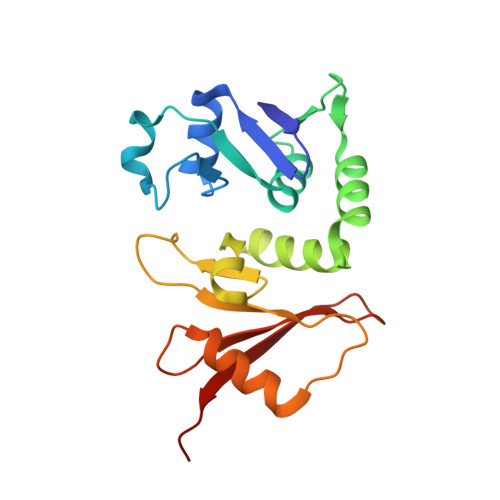
Functional importance of crenarchaea-specific extra-loop revealed by an X-ray structure of a heterotetrameric crenarchaeal splicing endonuclease
Yoshinari, S., Shiba, T., Inaoka, D.K., Itoh, T., Kurisu, G., Harada, S., Kita, K., Watanabe, Y.(2009) Nucleic Acids Res 37: 4787-4798
- PubMed: 19515941 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkp506
- Primary Citation Related Structures:
2ZYZ - PubMed Abstract:
Archaeal splicing endonucleases (EndAs) are currently classified into three groups. Two groups require a single subunit protein to form a homodimer or homotetramer. The third group requires two nonidentical protein components for the activity. To elucidate the molecular architecture of the two-subunit EndA system, we studied a crenarchaeal splicing endonuclease from Pyrobaculum aerophilum. In the present study, we solved a crystal structure of the enzyme at 1.7-A resolution. The enzyme adopts a heterotetrameric form composed of two catalytic and two structural subunits. By connecting the structural and the catalytic subunits of the heterotetrameric EndA, we could convert the enzyme to a homodimer that maintains the broad substrate specificity that is one of the characteristics of heterotetrameric EndA. Meanwhile, a deletion of six amino acids in a Crenarchaea-specific loop abolished the endonuclease activity even on a substrate with canonical BHB motif. These results indicate that the subunit architecture is not a major factor responsible for the difference of substrate specificity between single- and two-subunit EndA systems. Rather, the structural basis for the broad substrate specificity is built into the crenarchaeal splicing endonuclease itself.
- Department of Biomedical Chemistry, Graduate School of Medicine, The University of Tokyo, Hongo, Bunkyo-ku, Tokyo 113-0033, Japan. yoshinas@m.u-tokyo.ac.jp
Organizational Affiliation: