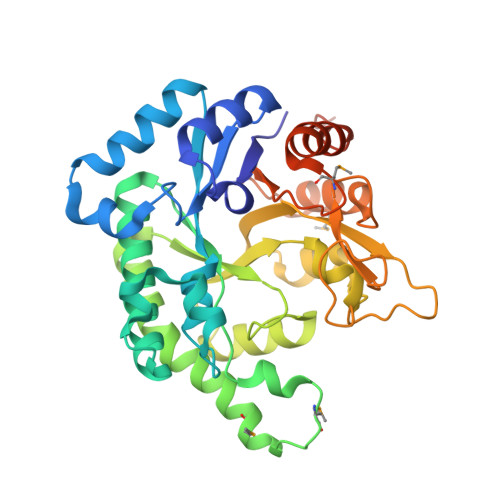
Crystal structure of SCO6571 from Streptomyces coelicolor A3(2).
Begum, P., Sakai, N., Hayashi, T., Gao, Y.G., Tamura, T., Watanabe, N., Yao, M., Tanaka, I.(2008) Protein Pept Lett 15: 709-712
- PubMed: 18782066
- DOI: https://doi.org/10.2174/092986608785133636
- Primary Citation of Related Structures:
2ZDS - PubMed Abstract:
SCO6571 protein from Streptomyces coelicolor A3(2) was overexpressed and purified using Rhodococcus erythropolis as an expressing host. Crystals of selenomethionine-substituted SCO6571 have been obtained by vapor diffusion method. SCO6571 crystals diffract to 2.3 A and were found to belong to the orthorhombic space group P2(1)2(1)2(1) with unit cell parameters a = 84.5, b = 171.6, c = 184.8 A. Six molecules in the asymmetric unit give a crystal volume per protein mass (V(M)) of 2.97 A (3) Da(-1) and solvent content of 58.6 %. The structure was solved by the single wavelength anomalous diffraction (SAD) method. SCO6571 is a TIM-barrel fold protein that assembles into a hexameric molecule with D(3) symmetry.
- Faculty of Advanced Life Sciences, Hokkaido University, Sapporo 060-0810, Japan.
Organizational Affiliation: