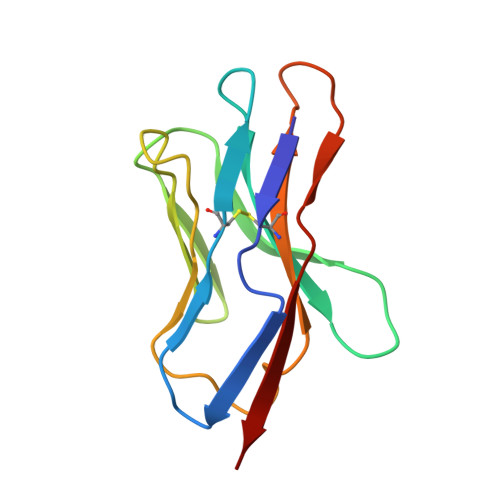
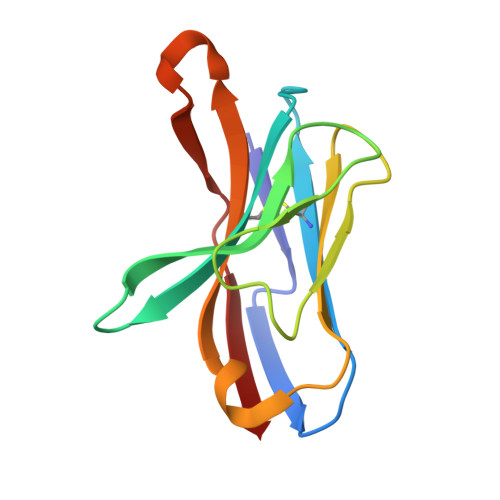
Structural evidence for a germline-encoded T cell receptor-major histocompatibility complex interaction 'codon'
Feng, D., Bond, C.J., Ely, L.K., Maynard, J., Garcia, K.C.(2007) Nat Immunol 8: 975-983
- PubMed: 17694060 Search on PubMed
- DOI: https://doi.org/10.1038/ni1502
- Primary Citation Related Structures:
2PXY, 2Z31, 2Z35 - PubMed Abstract:
All complexes of T cell receptors (TCRs) bound to peptide-major histocompatibility complex (pMHC) molecules assume a stereotyped binding 'polarity', despite wide variations in TCR-pMHC docking angles. However, existing TCR-pMHC crystal structures have failed to show broadly conserved pairwise interaction motifs. Here we determined the crystal structures of two TCRs encoded by the variable beta-chain 8.2 (V(beta)8.2), each bound to the MHC class II molecule I-A(u), and did energetic mapping of V(alpha) and V(beta) contacts with I-A(u). Together with two previously solved structures of V(beta)8.2-containing TCR-MHC complexes, we found four TCR-I-A complexes with structurally superimposable interactions between the V(beta) loops and the I-A alpha-helix. This examination of a narrow 'slice' of the TCR-MHC repertoire demonstrates what is probably one of many germline-derived TCR-MHC interaction 'codons'.
- Howard Hughes Medical Institute, Department of Molecular & Cellular Physiology, Stanford University School of Medicine, Stanford, California 94305, USA.
Organizational Affiliation: