Agonist-Bound Adenosine A(2A) Receptor Structures Reveal Common Features of Gpcr Activation.
Lebon, G., Warne, T., Edwards, P.C., Bennett, K., Langmead, C.J., Leslie, A.G.W., Tate, C.G.(2011) Nature 474: 521
- PubMed: 21593763
- DOI: https://doi.org/10.1038/nature10136
- Primary Citation Related Structures:
2YDO, 2YDV - PubMed Abstract:
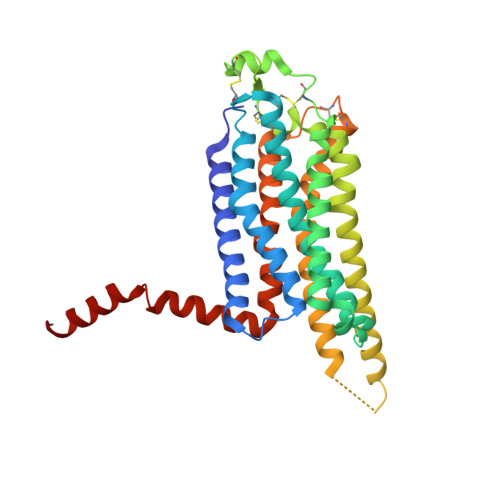
Adenosine receptors and β-adrenoceptors are G-protein-coupled receptors (GPCRs) that activate intracellular G proteins on binding the agonists adenosine or noradrenaline, respectively. GPCRs have similar structures consisting of seven transmembrane helices that contain well-conserved sequence motifs, indicating that they are probably activated by a common mechanism. Recent structures of β-adrenoceptors highlight residues in transmembrane region 5 that initially bind specifically to agonists rather than to antagonists, indicating that these residues have an important role in agonist-induced activation of receptors. Here we present two crystal structures of the thermostabilized human adenosine A(2A) receptor (A(2A)R-GL31) bound to its endogenous agonist adenosine and the synthetic agonist NECA. The structures represent an intermediate conformation between the inactive and active states, because they share all the features of GPCRs that are thought to be in a fully activated state, except that the cytoplasmic end of transmembrane helix 6 partially occludes the G-protein-binding site. The adenine substituent of the agonists binds in a similar fashion to the chemically related region of the inverse agonist ZM241385 (ref. 8). Both agonists contain a ribose group, not found in ZM241385, which extends deep into the ligand-binding pocket where it makes polar interactions with conserved residues in H7 (Ser 277(7.42) and His 278(7.43); superscripts refer to Ballesteros-Weinstein numbering) and non-polar interactions with residues in H3. In contrast, the inverse agonist ZM241385 does not interact with any of these residues and comparison with the agonist-bound structures indicates that ZM241385 sterically prevents the conformational change in H5 and therefore it acts as an inverse agonist. Comparison of the agonist-bound structures of A(2A)R with the agonist-bound structures of β-adrenoceptors indicates that the contraction of the ligand-binding pocket caused by the inward motion of helices 3, 5 and 7 may be a common feature in the activation of all GPCRs.
- MRC Laboratory of Molecular Biology, Hills Road, Cambridge CB2 0QH, UK.
Organizational Affiliation: