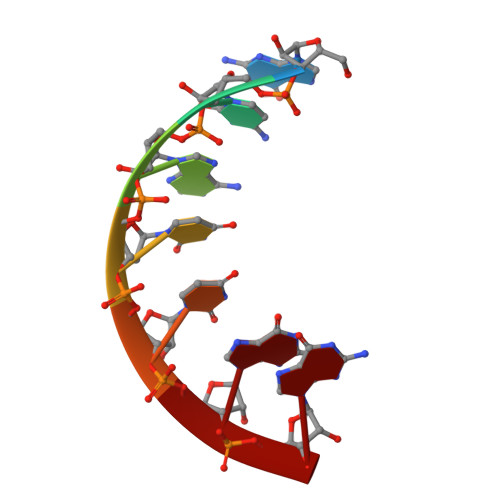
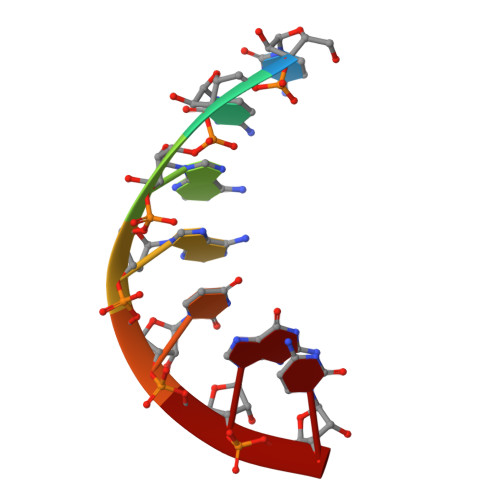
Crystal Structure of the Human Trnagly Microhelix Isoacceptor G9990 at 1.18 A Resolution
Eichert, A., Perbandt, M., Schreiber, A., Fuerste, J.P., Betzel, C., Erdmann, V.A., Foerster, C.(2009) Biochem Biophys Res Commun 380: 503
- PubMed: 19284994 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2009.01.127
- Primary Citation Related Structures:
2VUQ - PubMed Abstract:
The tRNA(Gly)/Glycyl-tRNA synthetase system belongs to the so called 'class II' in which tRNA identity elements consist of relative few and simple motifs, as compared to 'class I' where the tRNA determinants are more complicated and spread over different parts of the tRNA, mostly including the anticodon. The determinants from 'class II' although, are located in the aminoacyl stem and sometimes include the discriminator base. There exist predominant structure differences for the Glycyl-tRNA-synthetases and for the tRNA(Gly) identity elements comparing eucaryotic/archaebacterial and eubacterial systems. We focus on comparative X-ray structure analysis of tRNA(Gly) acceptor stem microhelices from different organisms. Here, we report the X-ray structure of the human tRNA(Gly) microhelix isoacceptor G9990 at 1.18A resolution. Superposition experiments to another human tRNA(Gly) microhelix and a detailed comparison of the RNA hydration patterns show a great number of water molecules with identical positions in both RNAs. This is the first structure comparison of hydration layers from two isoacceptor tRNA microhelices with a naturally occurring base pair exchange.
- Institute of Chemistry and Biochemistry, Free University Berlin, Thielallee 63, 14195 Berlin, Germany.
Organizational Affiliation: