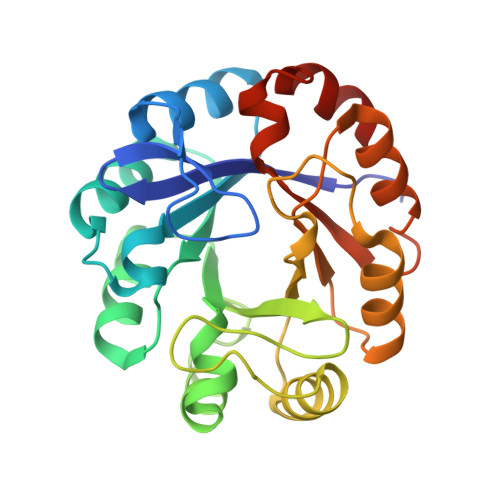
The Structure/Function Relationship of a Dual Substrate (Betaalpha)(8)-Isomerase
Wright, H., Noda-Garcia, L., Ochoa-Leyva, A., Hodgson, D.A., Fulop, V., Barona-Gomez, F.(2008) Biochem Biophys Res Commun 365: 16
- PubMed: 17967415 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2007.10.101
- Primary Citation Related Structures:
2VEP - PubMed Abstract:
Two structures of phosphoribosyl isomerase A (PriA) from Streptomyces coelicolor, involved in both histidine and tryptophan biosynthesis, were solved at 1.8A resolution. A closed conformer was obtained, which represents the first complete structure of PriA, revealing hitherto unnoticed molecular interactions and the occurrence of conformational changes. Inspection of these conformers, including ligand-docking simulations, allowed identification of residues involved in substrate recognition, chemical catalysis and conformational changes. These predictions were validated by mutagenesis and functional analysis. Arg19 and Ser81 were shown to play critical roles within the carboxyl and amino phosphate-binding sites, respectively; the catalytic residues Asp11 and Asp130 are responsible for both activities; and Thr166 and Asp171, which make an unusual contact, are likely to elicit the conformational changes needed for adopting the active site architectures. This represents the first report of the structure/function relationship of this (betaalpha)8-isomerase.
- Department of Biological Sciences, University of Warwick, Gibbet Hill Road, Coventry CV4 7AL, UK.
Organizational Affiliation: