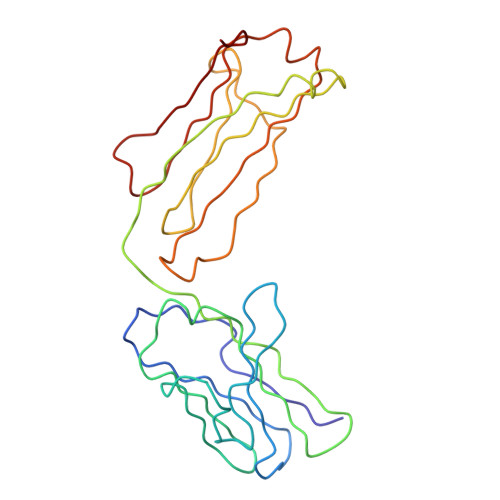
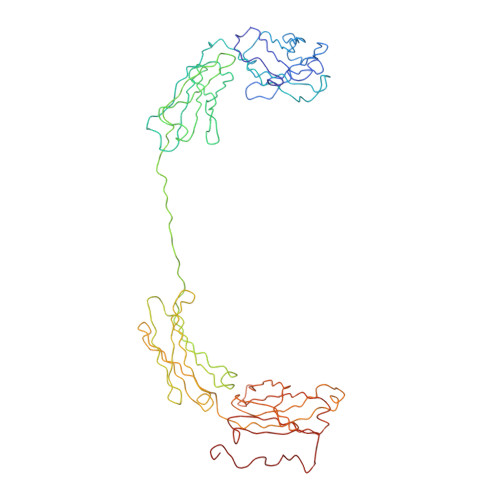
Implications of the near-planar solution structure of human myeloma dimeric IgA1 for mucosal immunity and IgA nephropathy
Bonner, A., Furtado, P.B., Almogren, A., Kerr, M.A., Perkins, S.J.(2008) J Immunol 180: 1008-1018
- PubMed: 18178841 Search on PubMed
- DOI: https://doi.org/10.4049/jimmunol.180.2.1008
- Primary Citation Related Structures:
2QTJ - PubMed Abstract:
IgA is unique in being able to form a diverse range of polymeric structures. Increases in the levels of dimeric IgA1 (dIgA1) in serum have been implicated in diseases such as IgA nephropathy. We have determined the solution structure for dIgA1 by synchrotron x-ray and neutron scattering and analytical ultracentrifugation. The Guinier radius of gyration (RG) of 7.60-8.65 nm indicated that the two monomers within dIgA1 are arranged in an extended conformation. The distance distribution curve P(r) gave an overall length (L) of 22-26 nm. These results were confirmed by the sedimentation coefficient and frictional ratio of dIgA1. Constrained scattering modeling starting from the IgA1 monomer solution structure revealed a near-planar dimer structure for dIgA1. The two Fc regions form a slightly bent arrangement in which they form end-to-end contacts, and the J chain was located at this interface. This structure was refined by optimizing the position of the four Fab regions. From this, the best-fit solution structures show that the four Fab Ag-binding sites are independent of one another, and the two Fc regions are accessible to receptor binding. This arrangement allows dIgA1 to initiate specific immune responses by binding to FcalphaRI receptors, while still retaining Ag-binding ability, and to be selectively transported to mucosal surfaces by binding to the polymeric Ig receptor to form secretory IgA. A mechanism for the involvement of dIgA1 oligomers in the pathology of IgA nephropathy is discussed in the light of this near-planar structure.
- Department of Biochemistry and Molecular Biology, University College London, London, United Kingdom.
Organizational Affiliation: