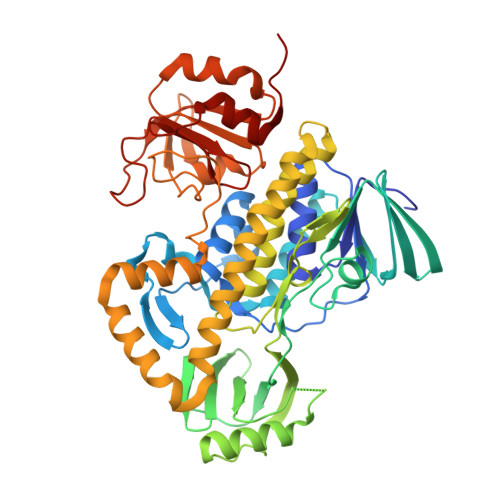
Crystal structures of two aromatic hydroxylases involved in the early tailoring steps of angucycline biosynthesis
Koskiniemi, H., Metsa-Ketela, M., Dobritzsch, D., Kallio, P., Korhonen, H., Mantsala, P., Schneider, G., Niemi, J.(2007) J Mol Biology 372: 633-648
- PubMed: 17669423 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2007.06.087
- Primary Citation Related Structures:
2QA1, 2QA2 - PubMed Abstract:
Angucyclines are aromatic polyketides produced in Streptomycetes via complex enzymatic biosynthetic pathways. PgaE and CabE from S. sp PGA64 and S. sp. H021 are two related homo-dimeric FAD and NADPH dependent aromatic hydroxylases involved in the early steps of the angucycline core modification. Here we report the three-dimensional structures of these two enzymes determined by X-ray crystallography using multiple anomalous diffraction and molecular replacement, respectively, to resolutions of 1.8 A and 2.7 A. The enzyme subunits are built up of three domains, a FAD binding domain, a domain involved in substrate binding and a C-terminal thioredoxin-like domain of unknown function. The structure analysis identifies PgaE and CabE as members of the para-hydroxybenzoate hydroxylase (pHBH) fold family of aromatic hydroxylases. In contrast to phenol hydroxylase and 3-hydroxybenzoate hydroxylase that utilize the C-terminal domain for dimer formation, this domain is not part of the subunit-subunit interface in PgaE and CabE. Instead, dimer assembly occurs through interactions of their FAD binding domains. FAD is bound non-covalently in the "in"-conformation. The active sites in the two enzymes differ significantly from those of other aromatic hydroxylases. The volumes of the active site are significantly larger, as expected in view of the voluminous tetracyclic angucycline substrates. The structures further suggest that substrate binding and catalysis may involve dynamic rearrangements of the middle domain relative to the other two domains. Site-directed mutagenesis studies of putative catalytic groups in the active site of PgaE argue against enzyme-catalyzed substrate deprotonation as a step in catalysis. This is in contrast to pHBH, where deprotonation/protonation of the substrate has been suggested as an essential part of the enzymatic mechanism.
- Department of Medical Biochemistry and Biophysics, Karolinska Institutet, S-171 77 Stockholm, Sweden.
Organizational Affiliation: