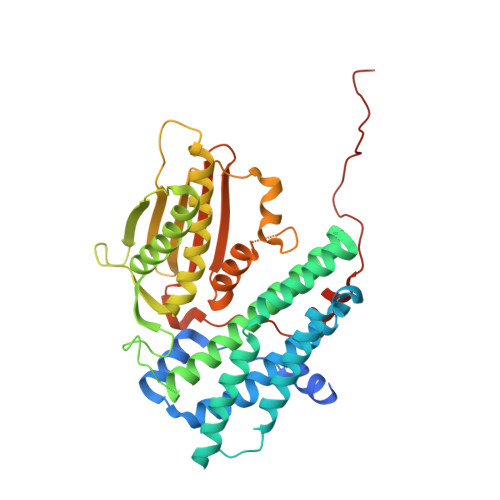
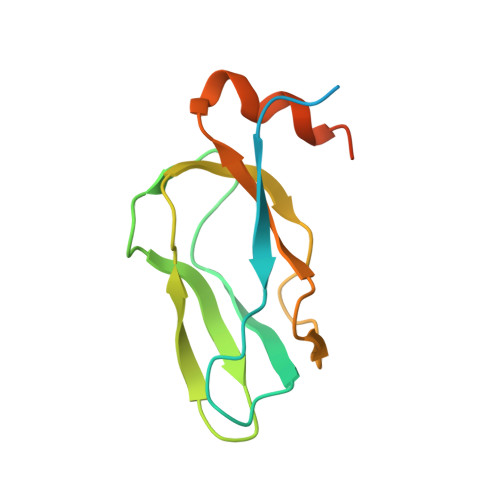
Distinct Structural Mechanisms for Inhibition of Pyruvate Dehydrogenase Kinase Isoforms by AZD7545, Dichloroacetate, and Radicicol.
Kato, M., Li, J., Chuang, J.L., Chuang, D.T.(2007) Structure 15: 992-1004
- PubMed: 17683942 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2007.07.001
- Primary Citation Related Structures:
2Q8F, 2Q8G, 2Q8H, 2Q8I - PubMed Abstract:
Pyruvate dehydrogenase kinase (PDK) isoforms are molecular switches that downregulate the pyruvate dehydrogenase complex (PDC) by reversible phosphorylation in mitochondria. We have determined structures of human PDK1 or PDK3 bound to the inhibitors AZD7545, dichloroacetate (DCA), and radicicol. We show that the trifluoromethylpropanamide end of AZD7545 projects into the lipoyl-binding pocket of PDK1. This interaction results in inhibition of PDK1 and PDK3 activities by aborting kinase binding to the PDC scaffold. Paradoxically, AZD7545 at saturating concentrations robustly increases scaffold-free PDK3 activity, similar to the inner lipoyl domain. Good DCA density is present in the helix bundle in the N-terminal domain of PDK1. Bound DCA promotes local conformational changes that are communicated to both nucleotide-binding and lipoyl-binding pockets of PDK1, leading to the inactivation of kinase activity. Finally, radicicol inhibits kinase activity by binding directly to the ATP-binding pocket of PDK3, similar to Hsp90 and Topo VI from the same ATPase/kinase superfamily.
- Department of Internal Medicine, University of Texas Southwestern Medical Center, Dallas, TX 75390, USA.
Organizational Affiliation: