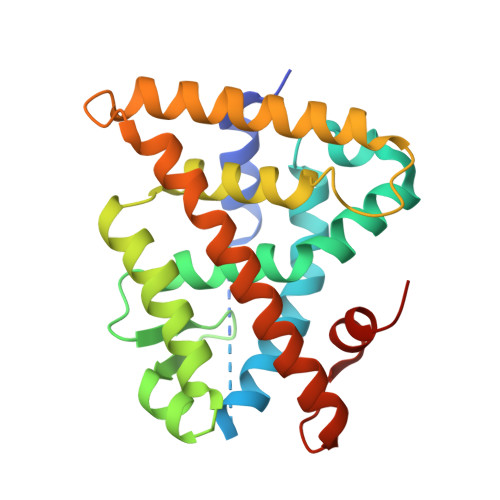
Modulators of the structural dynamics of the retinoid X receptor to reveal receptor function.
Nahoum, V., Perez, E., Germain, P., Rodriguez-Barrios, F., Manzo, F., Kammerer, S., Lemaire, G., Hirsch, O., Royer, C.A., Gronemeyer, H., de Lera, A.R., Bourguet, W.(2007) Proc Natl Acad Sci U S A 104: 17323-17328
- PubMed: 17947383 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0705356104
- Primary Citation Related Structures:
2P1T, 2P1U, 2P1V - PubMed Abstract:
Retinoid X receptors (RXRalpha, -beta, and -gamma) occupy a central position in the nuclear receptor superfamily, because they form heterodimers with many other family members and hence are involved in the control of a variety of (patho)physiologic processes. Selective RXR ligands, referred to as rexinoids, are already used or are being developed for cancer therapy and have promise for the treatment of metabolic diseases. However, important side effects remain associated with existing rexinoids. Here we describe the rational design and functional characterization of a spectrum of RXR modulators ranging from partial to pure antagonists and demonstrate their utility as tools to probe the implication of RXRs in cell biological phenomena. One of these ligands renders RXR activity particularly sensitive to coactivator levels and has the potential to act as a cell-specific RXR modulator. A combination of crystallographic and fluorescence anisotropy studies reveals the molecular details accounting for the agonist-to-antagonist transition and provides direct experimental evidence for a correlation between the pharmacological activity of a ligand and its impact on the structural dynamics of the activation helix H12. Using RXR and its cognate ligands as a model system, our correlative analysis of 3D structures and dynamic data provides an original view on ligand actions and enables the establishment of mechanistic concepts, which will aid in the development of selective nuclear receptor modulators.
- Institut National de la Santé et de la Recherche Médicale, U554, 34090 Montpellier, France.
Organizational Affiliation: