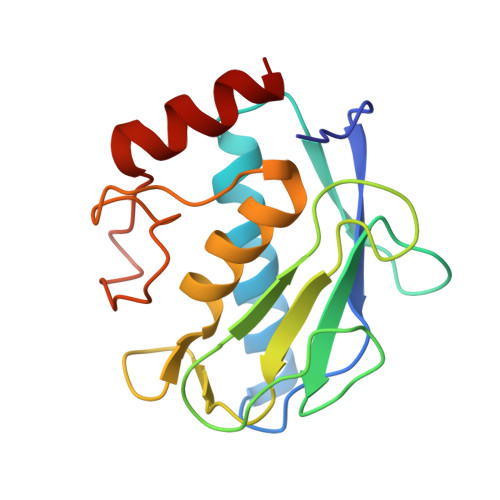
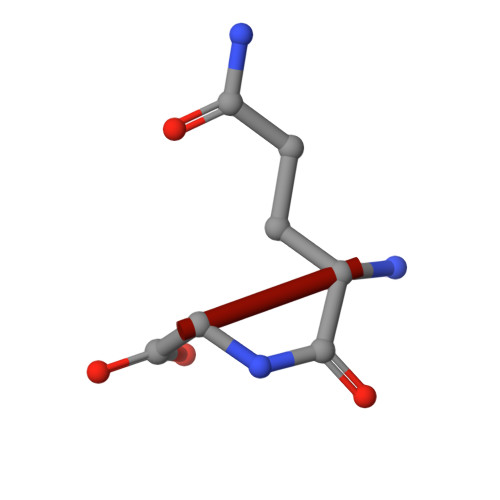
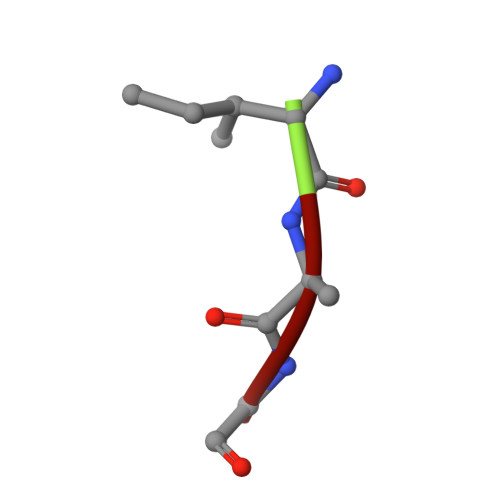
Snapshots of the reaction mechanism of matrix metalloproteinases.
Bertini, I., Calderone, V., Fragai, M., Luchinat, C., Maletta, M., Yeo, K.J.(2006) Angew Chem Int Ed Engl 45: 7952-7955
- PubMed: 17096442 Search on PubMed
- DOI: https://doi.org/10.1002/anie.200603100
- Primary Citation Related Structures:
2OXU, 2OXW, 2OXZ, 2OY2, 2OY4 - Magnetic Resonance Center (CERM), University of Florence, Via L. Sacconi 6, 50019 Sesto Fiorentino, Italy. ivanobertini@cerm.unifi.it
Organizational Affiliation: