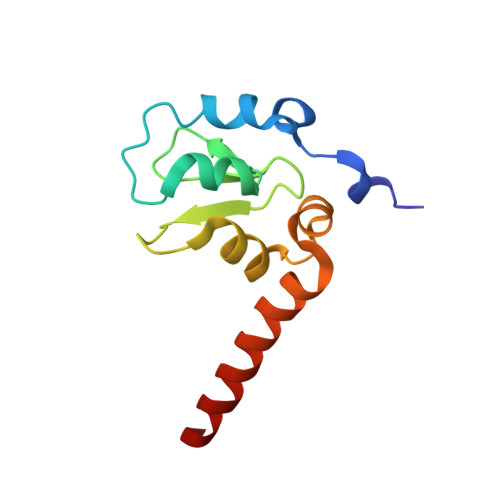
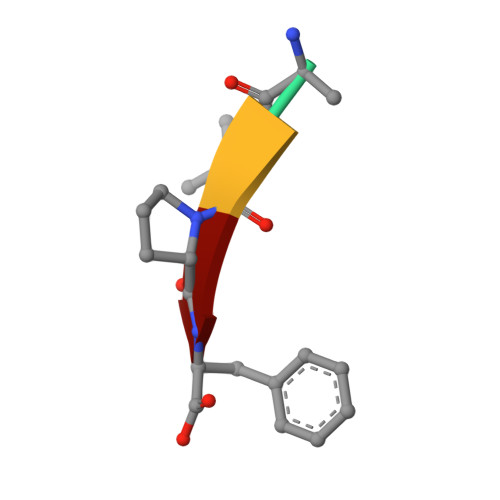
Structure-activity based study of the Smac-binding pocket within the BIR3 domain of XIAP.
Wist, A.D., Gu, L., Riedl, S.J., Shi, Y., McLendon, G.L.(2007) Bioorg Med Chem 15: 2935-2943
- PubMed: 17336535 Search on PubMed
- DOI: https://doi.org/10.1016/j.bmc.2007.02.010
- Primary Citation Related Structures:
2OPY, 2OPZ - PubMed Abstract:
A small series of peptide mimics was designed and synthesized to contain a heterocyclic ring in place of the potentially labile N-terminal peptide bond of the tetrapeptide containing the Smac-XIAP-binding motif. Two Smac mimics were shown to bind to the BIR3 domain of XIAP with moderate affinity and one displayed increased activity in cells relative to the Smac peptides. The structures of BIR3-XIAP in complex with a Smac peptide and a peptide mimic were solved and analyzed to elucidate the structure-activity relationship surrounding the Smac-binding domain within BIR3-XIAP.
- Department of Chemistry, Princeton University, Princeton, NJ 08544, USA. aislyn.wist@mssm.edu
Organizational Affiliation: