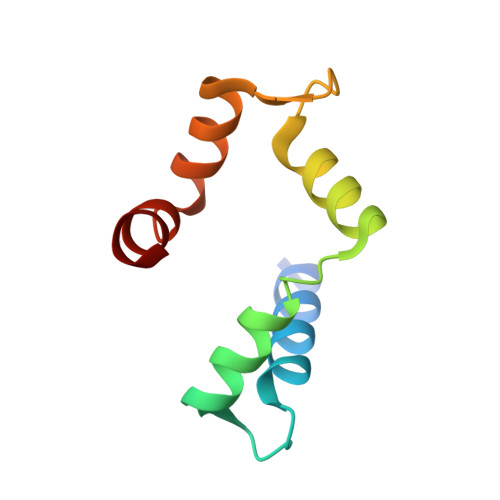
Three-dimensional structure of the cross-reactive pollen allergen Che a 3: visualizing cross-reactivity on the molecular surfaces of weed, grass, and tree pollen allergens.
Verdino, P., Barderas, R., Villalba, M., Westritschnig, K., Valenta, R., Rodriguez, R., Keller, W.(2008) J Immunol 180: 2313-2321
- PubMed: 18250440 Search on PubMed
- DOI: https://doi.org/10.4049/jimmunol.180.4.2313
- Primary Citation Related Structures:
2OPO - PubMed Abstract:
Two EF-hand calcium-binding allergens (polcalcins) occur in the pollen of a wide variety of unrelated plants as highly cross-reactive allergenic molecules. We report the expression, purification, immunological characterization, and the 1.75-A crystal structure of recombinant Che a 3 (rChe a 3), the polcalcin from the weed Chenopodium album. The three-dimensional structure of rChe a 3 resembles an alpha-helical fold that is essentially identical with that of the two EF-hand allergens from birch pollen, Bet v 4, and timothy grass pollen, Phl p 7. The extensive cross-reactivity between Che a 3 and Phl p 7 is demonstrated by competition experiments with IgE Abs from allergic patients as well as specific Ab probes. Amino acid residues that are conserved for the two EF-hand allergen family were identified in multiple sequence alignments of polcalcins from 15 different plants. Next, the three-dimensional structures of rChe a 3, rPhl p 7, and rBet v 4 were used to identify conserved amino acids with high surface exposition to visualize surface patches as potential targets for the polyclonal IgE Ab response of allergic patients. The essentially identical three-dimensional structures of rChe a 3, rPhl p 7, and rBet v 4 explain the extensive cross-reactivity of allergic patients IgE Abs with two EF-hand allergens from unrelated plants. In addition, analyzing the three-dimensional structures of cross-reactive Ags for conserved and surface exposed amino acids may be a first approach to mapping the conformational epitopes on disease-related Ags that are recognized by polyclonal patient Abs.
- Institute of Chemistry, Structural Biology, University of Graz, Heinrichstrasse 28, Graz, Austria.
Organizational Affiliation: