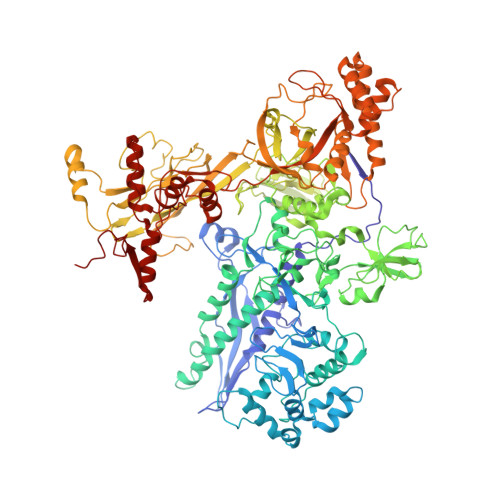
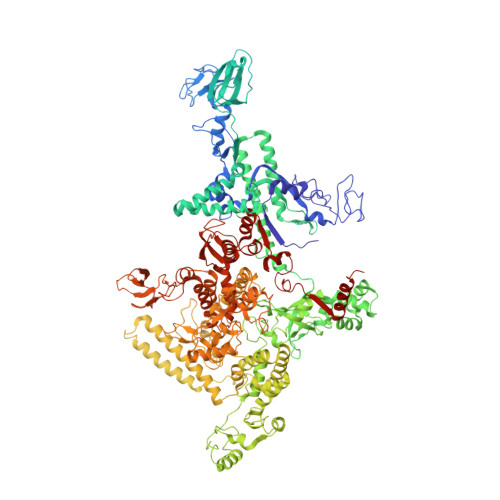
Structural basis for transcription elongation by bacterial RNA polymerase.
Vassylyev, D.G., Vassylyeva, M.N., Perederina, A., Tahirov, T.H., Artsimovitch, I.(2007) Nature 448: 157-162
- PubMed: 17581590 Search on PubMed
- DOI: https://doi.org/10.1038/nature05932
- Primary Citation Related Structures:
2O5I - PubMed Abstract:
The RNA polymerase elongation complex (EC) is both highly stable and processive, rapidly extending RNA chains for thousands of nucleotides. Understanding the mechanisms of elongation and its regulation requires detailed information about the structural organization of the EC. Here we report the 2.5-A resolution structure of the Thermus thermophilus EC; the structure reveals the post-translocated intermediate with the DNA template in the active site available for pairing with the substrate. DNA strand separation occurs one position downstream of the active site, implying that only one substrate at a time can specifically bind to the EC. The upstream edge of the RNA/DNA hybrid stacks on the beta'-subunit 'lid' loop, whereas the first displaced RNA base is trapped within a protein pocket, suggesting a mechanism for RNA displacement. The RNA is threaded through the RNA exit channel, where it adopts a conformation mimicking that of a single strand within a double helix, providing insight into a mechanism for hairpin-dependent pausing and termination.
- Department of Biochemistry and Molecular Genetics, University of Alabama at Birmingham, Schools of Medicine and Dentistry, 402B Kaul Genetics Building, 720 20th Street South, Birmingham, Alabama 35294, USA. dmitry@uab.edu
Organizational Affiliation: