Site-specific recombination of nitrogen-fixation genes in cyanobacteria by XisF-XisH-XisI complex: Structures and models.
Hwang, W.C., Golden, J.W., Pascual, J., Xu, D., Cheltsov, A., Godzik, A.(2014) Proteins
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
(2014) Proteins
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| FdxN element excision controlling factor XisI | 126 | Trichormus variabilis ATCC 29413 | Mutation(s): 0 Gene Names: YP_321976.1 | 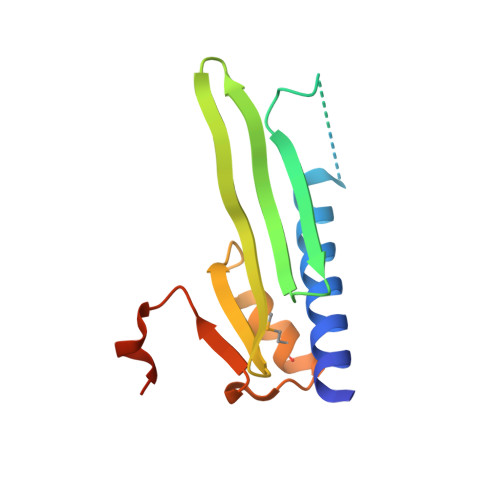 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q3MD55 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 122.44 | α = 90 |
| b = 122.44 | β = 90 |
| c = 77.076 | γ = 120 |
| Software Name | Purpose |
|---|---|
| MolProbity | model building |
| SHELX | phasing |
| REFMAC | refinement |
| SCALA | data scaling |
| PDB_EXTRACT | data extraction |
| MOSFLM | data reduction |
| CCP4 | data scaling |
| SHELXD | phasing |
| autoSHARP | phasing |