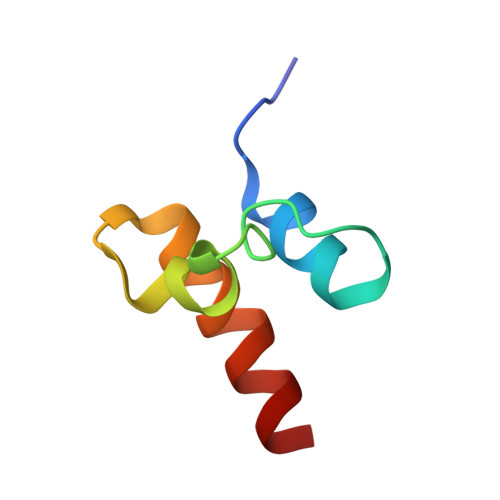
Unveiling the structural determinants of KIAA0323 binding preference for NEDD8.
Santonico, E., Nepravishta, R., Mattioni, A., Valentini, E., Mandaliti, W., Procopio, R., Iannuccelli, M., Castagnoli, L., Polo, S., Paci, M., Cesareni, G.To be published.