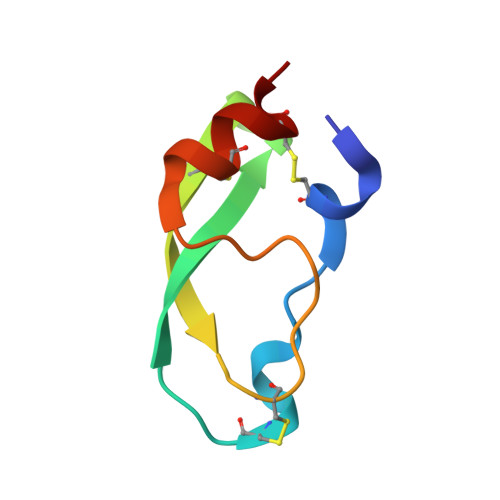
NMR solution structure of a Chymotrypsin inhibitor from the Taiwan cobra Naja naja atra.
Lin, Y.J., Ikeya, T., Guntert, P., Chang, L.S.(2013) Molecules 18: 8906-8918
- PubMed: 23896616 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.3390/molecules18088906
- Primary Citation Related Structures:
2M99 - PubMed Abstract:
The Taiwan cobra (Naja naja atra) chymotrypsin inhibitor (NACI) consists of 57 amino acids and is related to other Kunitz-type inhibitors such as bovine pancreatic trypsin inhibitor (BPTI) and Bungarus fasciatus fraction IX (BF9), another chymotrypsin inhibitor. Here we present the solution structure of NACI. We determined the NMR structure of NACI with a root-mean-square deviation of 0.37 Å for the backbone atoms and 0.73 Å for the heavy atoms on the basis of 1,075 upper distance limits derived from NOE peaks measured in its NOESY spectra. To investigate the structural characteristics of NACI, we compared the three-dimensional structure of NACI with BPTI and BF9. The structure of the NACI protein comprises one 310-helix, one α-helix and one double-stranded antiparallel β-sheet, which is comparable with the secondary structures in BPTI and BF9. The RMSD value between the mean structures is 1.09 Å between NACI and BPTI and 1.27 Å between NACI and BF9. In addition to similar secondary and tertiary structure, NACI might possess similar types of protein conformational fluctuations as reported in BPTI, such as Cys14-Cys38 disulfide bond isomerization, based on line broadening of resonances from residues which are mainly confined to a region around the Cys14-Cys38 disulfide bond.
- Graduate Institute of Natural Products and Center of Excellence for Environmental Medicine, Kaohsiung Medical University, No.100, Shi-Chuan 1st Road, San-Ming District, Kaohsiung 807, Taiwan. yjlin@kmu.edu.tw
Organizational Affiliation: