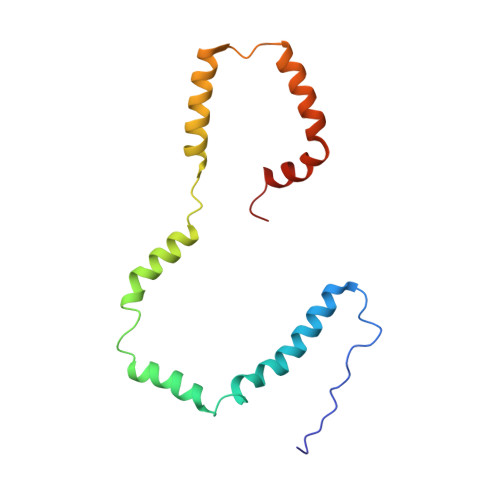
Structural Insights of tBid, the Caspase-8-activated Bid, and Its BH3 Domain.
Wang, Y., Tjandra, N.(2013) J Biological Chem 288: 35840-35851
- PubMed: 24158446 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M113.503680
- Primary Citation Related Structures:
2M5I - PubMed Abstract:
The Bcl-2 family proteins regulate mitochondria-mediated apoptosis through intricate molecular mechanisms. One of the pro-apoptotic proteins, tBid, can induce apoptosis by promoting Bax activation, Bax homo-oligomerization, and mitochondrial outer membrane permeabilization. Association of tBid on the mitochondrial outer membrane is key to its biological function. Therefore knowing the conformation of tBid on the membrane will be the first step toward understanding its crucial role in triggering apoptosis. Here, we present NMR characterization of the structure and dynamics of human tBid in 1-palmitoyl-2-hydroxy-sn-glycero-3-[phospho-RAC-(1-glycerol)] micelles. Our data showed that tBid is monomeric with six well defined α-helices in the micelles. Compared with the full-length Bid structure, a longer flexible loop between tBid helix α4 and α5 was observed. Helices in tBid do not pack into a compact-fold but form an extended structure with a C-shape configuration in the micelles. All six tBid helices were shown to interact with LPPG micelles, with helix α6 and α7 being more embedded. Of note, the BH3-containing helix α3, which was previously believed to be exposed above the membrane surface, is also membrane associated, suggesting an "on the membrane" binding mode for tBid interaction with Bax. Our data provided structural details on the membrane-associated state of tBid and the functional implications of its membrane-associated BH3 domain.
- From the Laboratory of Molecular Biophysics, Biochemistry and Biophysics Center, NHLBI, National Institutes of Health, Bethesda, Maryland 20892.
Organizational Affiliation: