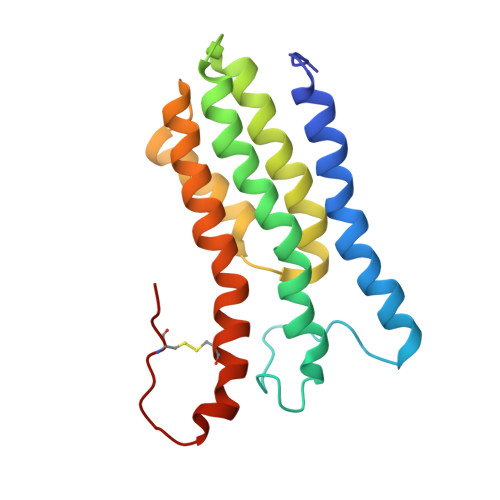
Solution structure of decorin-binding protein A from Borrelia burgdorferi.
Wang, X.(2012) Biochemistry 51: 8353-8362
- PubMed: 22985470
- DOI: https://doi.org/10.1021/bi3007093
- Primary Citation of Related Structures:
2LQU - PubMed Abstract:
Decorin-binding protein A (DBPA) is an important lipoprotein from the bacterium Borrelia burgdorferi, the causative agent of Lyme disease. The absence of DBPA drastically reduces the pathogenic potential of the bacterium, and biochemical evidence indicates DBPA's interactions with the glycosaminoglycan (GAG) portion of decorin are crucial to its function. We have determined the solution structure of DBPA and studied DBPA's interactions with various forms of GAGs. DBPA is determined to be a helical bundle protein consisting of five helices held together by a strong hydrophobic core. The structure also possesses a basic patch formed by portions of two helices and two flexible linkers. Low-molecular mass heparin-induced chemical shift perturbations for residues in the region as well as increases in signal intensities of select residues in their presence confirm residues in the pocket are perturbed by heparin binding. Dermatan sulfate fragments, the dominant GAG type found on decorin, were shown to have lower affinity than heparin but are still capable of binding DBPA.
- Department of Chemistry and Biochemistry, Arizona State University, Tempe, AZ 85287, USA. xuwang@asu.edu
Organizational Affiliation: