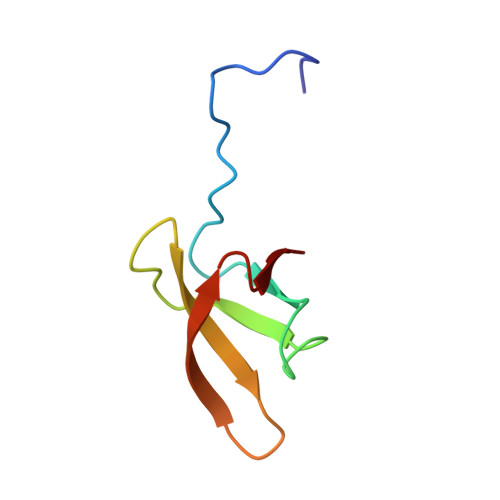
An alpha helix to beta barrel domain switch transforms the transcription factor RfaH into a translation factor.
Burmann, B.M., Knauer, S.H., Sevostyanova, A., Schweimer, K., Mooney, R.A., Landick, R., Artsimovitch, I., Rosch, P.(2012) Cell 150: 291-303
- PubMed: 22817892 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.cell.2012.05.042
- Primary Citation Related Structures:
2LCL - PubMed Abstract:
NusG homologs regulate transcription and coupled processes in all living organisms. The Escherichia coli (E. coli) two-domain paralogs NusG and RfaH have conformationally identical N-terminal domains (NTDs) but dramatically different carboxy-terminal domains (CTDs), a β barrel in NusG and an α hairpin in RfaH. Both NTDs interact with elongating RNA polymerase (RNAP) to reduce pausing. In NusG, NTD and CTD are completely independent, and NusG-CTD interacts with termination factor Rho or ribosomal protein S10. In contrast, RfaH-CTD makes extensive contacts with RfaH-NTD to mask an RNAP-binding site therein. Upon RfaH interaction with its DNA target, the operon polarity suppressor (ops) DNA, RfaH-CTD is released, allowing RfaH-NTD to bind to RNAP. Here, we show that the released RfaH-CTD completely refolds from an all-α to an all-β conformation identical to that of NusG-CTD. As a consequence, RfaH-CTD binding to S10 is enabled and translation of RfaH-controlled operons is strongly potentiated. PAPERFLICK:
- Lehrstuhl Biopolymere und Forschungszentrum für Bio-Makromoleküle, Universität Bayreuth, Universitätsstraße 30, 95447 Bayreuth, Germany.
Organizational Affiliation: