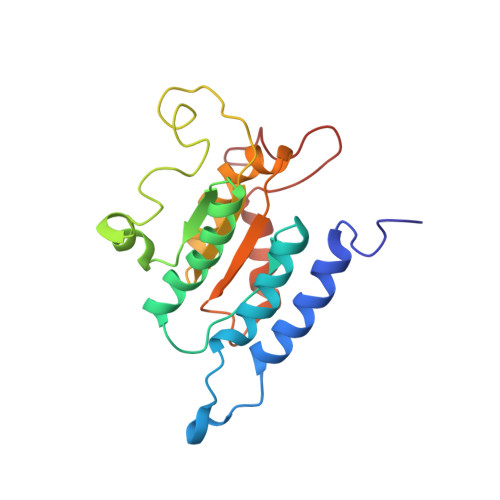
Solution structure and small angle scattering analysis of TraI (381-569).
Wright, N.T., Raththagala, M., Hemmis, C.W., Edwards, S., Curtis, J.E., Krueger, S., Schildbach, J.F.(2012) Proteins 80: 2250-2261
- PubMed: 22611034 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/prot.24114
- Primary Citation Related Structures:
2L8B - PubMed Abstract:
TraI, the F plasmid-encoded nickase, is a 1756 amino acid protein essential for conjugative transfer of plasmid DNA from one bacterium to another. Although crystal structures of N- and C-terminal domains of F TraI have been determined, central domains of the protein are structurally unexplored. The central region (between residues 306 and 1520) is known to both bind single-stranded DNA (ssDNA) and unwind DNA through a highly processive helicase activity. Here, we show that the ssDNA binding site is located between residues 381 and 858, and we also present the high-resolution solution structure of the N-terminus of this region (residues 381-569). This fragment folds into a four-strand parallel β sheet surrounded by α helices, and it resembles the structure of the N-terminus of helicases such as RecD and RecQ despite little sequence similarity. The structure supports the model that F TraI resulted from duplication of a RecD-like domain and subsequent specialization of domains into the more N-terminal ssDNA binding domain and the more C-terminal domain containing helicase motifs. In addition, we provide evidence that the nickase and ssDNA binding domains of TraI are held close together by an 80-residue linker sequence that connects the two domains. These results suggest a possible physical explanation for the apparent negative cooperativity between the nickase and ssDNA binding domain.
- Department of Biology, Johns Hopkins University, Baltimore, Maryland 21218, USA.
Organizational Affiliation: