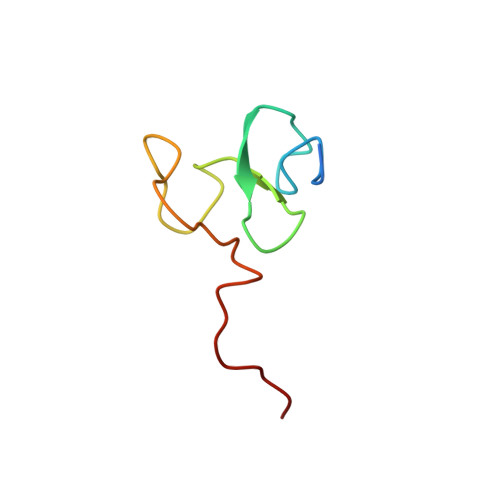
Plant Homeodomain (PHD) Fingers of CHD4 Are Histone H3-binding Modules with Preference for Unmodified H3K4 and Methylated H3K9
Mansfield, R.E., Musselman, C.A., Kwan, A.H., Oliver, S.S., Garske, A.L., Davrazou, F., Denu, J.M., Kutateladze, T.G., Mackay, J.P.(2011) J Biological Chem 286: 11779-11791
- PubMed: 21278251 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M110.208207
- Primary Citation Related Structures:
2L5U, 2L75 - PubMed Abstract:
A major challenge in chromatin biology is to understand the mechanisms by which chromatin is remodeled into active or inactive states as required during development and cell differentiation. One complex implicated in these processes is the nucleosome remodeling and histone deacetylase (NuRD) complex, which contains both histone deacetylase and nucleosome remodeling activities and has been implicated in the silencing of subsets of genes involved in various stages of cellular development. Chromodomain-helicase-DNA-binding protein 4 (CHD4) is a core component of the NuRD complex and contains a nucleosome remodeling ATPase domain along with two chromodomains and two plant homeodomain (PHD) fingers. We have previously demonstrated that the second PHD finger of CHD4 binds peptides corresponding to the N terminus of histone H3 methylated at Lys(9). Here, we determine the solution structure of PHD2 in complex with H3K9me3, revealing the molecular basis of histone recognition, including a cation-π recognition mechanism for methylated Lys(9). Additionally, we demonstrate that the first PHD finger also exhibits binding to the N terminus of H3, and we establish the histone-binding surface of this domain. This is the first instance where histone binding ability has been demonstrated for two separate PHD modules within the one protein. These findings suggest that CHD4 could bind to two H3 N-terminal tails on the same nucleosome or on two separate nucleosomes simultaneously, presenting exciting implications for the mechanism by which CHD4 and the NuRD complex could direct chromatin remodeling.
- School of Molecular Bioscience, University of Sydney, Sydney, New South Wales, Australia.
Organizational Affiliation: