The structure of E-protein activation domain 1 bound to the KIX domain of CBP/p300 elucidates leukemia induction by E2A-PBX1
Denis, C.M., Chitayat, S., Plevin, M.J., Liu, S., Spencer, H.L., Ikura, M., LeBrun, D.P., Smith, S.P.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| CREB-binding protein | 87 | Homo sapiens | Mutation(s): 0 Gene Names: CREBBP, CBP EC: 2.3.1.48 (PDB Primary Data), 2.3.1 (UniProt) | 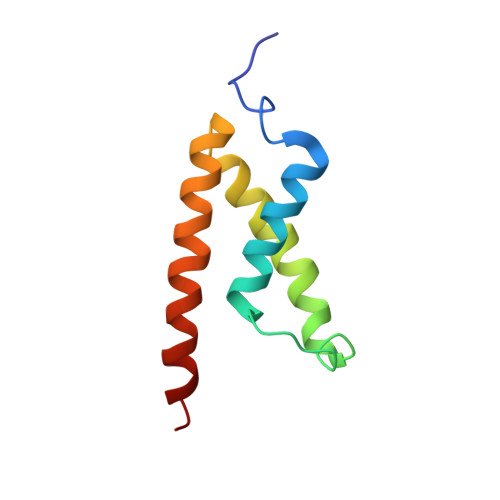 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q92793 GTEx: ENSG00000005339 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q92793 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Transcription factor 4 | 19 | Homo sapiens | Mutation(s): 0 Gene Names: TCF4, BHLHB19, ITF2, SEF2 | 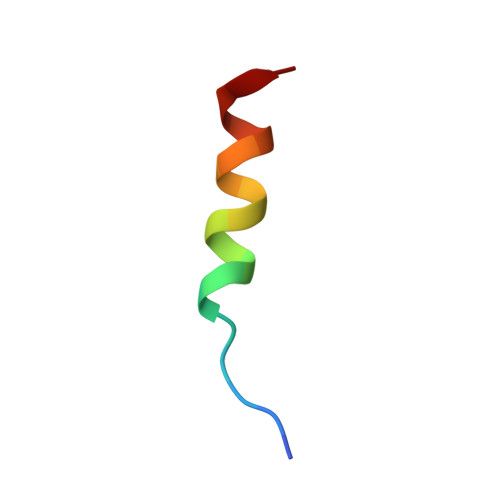 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P15884 GTEx: ENSG00000196628 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P15884 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||