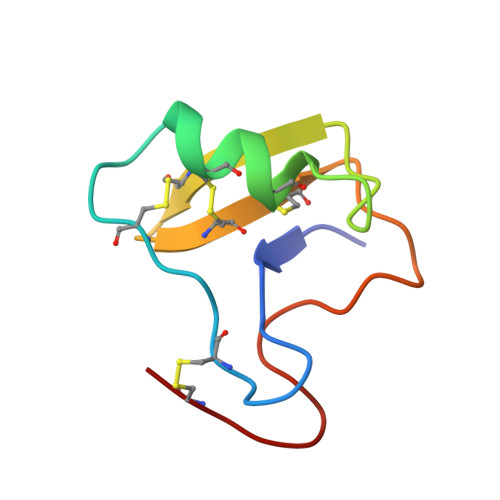
Solution structure of Cn5, a crustacean toxin found in the venom of the scorpions Centruroides noxius and Centruroides suffusus suffusus.
Corzo, G., Prochnicka-Chalufour, A., Garcia, B.I., Possani, L.D., Delepierre, M.(2009) Biochim Biophys Acta 1794: 1591-1598
- PubMed: 19631296 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbapap.2009.07.006
- Primary Citation Related Structures:
2KJA - PubMed Abstract:
The crustacean toxin Cn5 from Centruroides noxius Hoffmann and peptide Css39.8 from Centruroides suffusus suffusus scorpion venoms are identical peptides, as confirmed by amino acid sequence of purified toxins and by DNA sequencing of the two respective cloned genes. Therefore in this communication they will be simply named Cn5. Cn5 is a 66 amino acid long peptide with four disulfide bridges, formed between pairs of cysteines: C1-C8, C2-C5, C3-C6, and C4-C7 (the numbers indicate the relative positions of the cysteine residues in the primary structure). This peptide is non-toxic to mammals but deadly to arthropods (LD(50) 28.5 mg/g body weight of crayfish). Its three-dimensional structure was determined by NMR using a total of 965 meaningful distance constraints derived from the volume integration of the 2D NOESY spectra. The Cn5 structure displays a mixed alpha/beta fold stabilized by four disulfide bridges, with a kink induced by a cis-proline in its C-terminal part. Cn5 electrostatic surface is compared to that of Cn2 toxin toxic to mammals. The local differences produced by additional or substituted residues that would influence toxin selectivity towards mammalian or crustacean Na(+) channels are discussed.
- Departamento de Medicina Molecular y Bioprocesos, Instituto de Biotecnología, Universidad Nacional Autónoma de México, UNAM, Apartado Postal 510-3, Cuernavaca Morelos, 62210, Mexico.
Organizational Affiliation: