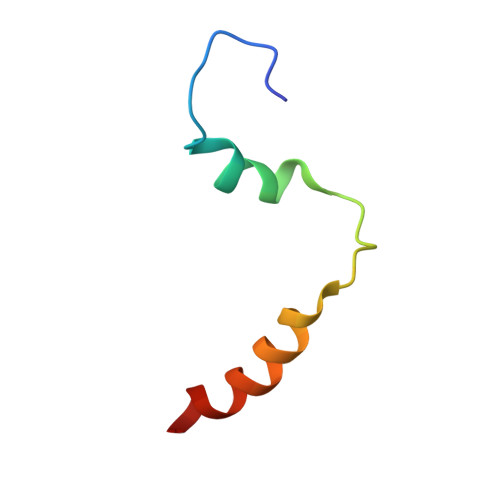
Functional and structural characterization of a dense core secretory granule sorting domain from the PC1/3 protease
Dikeakos, J.D., Di Lello, P., Lacombe, M.J., Ghirlando, R., Legault, P., Reudelhuber, T.L., Omichinski, J.G.(2009) Proc Natl Acad Sci U S A 106: 7408-7413
- PubMed: 19376969 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0809576106
- Primary Citation Related Structures:
2KDT, 2KE3 - PubMed Abstract:
Several peptide hormones are initially synthesized as inactive precursors. It is only on entry of these prohormones and their processing proteases into dense core secretory granules (DCSGs) that the precursors are cleaved to generate their active forms. Prohormone convertase (PC)1/3 is a processing protease that is targeted to DCSGs. The signal for targeting PC1/3 to DCSGs resides in its carboxy-terminal tail (PC1/3(617-753)), where 3 regions (PC1/3(617-625), PC1/3(665-682), and PC1/3(711-753)) are known to aid in sorting and membrane association. In this article, we have determined a high-resolution structure of the extreme carboxy-terminal sorting domain, PC1/3(711-753) in micelles by NMR spectroscopy. PC1/3(711-753) contains 2 alpha helices located between residues 722-728 and 738-750. Functional assays demonstrate that the second helix (PC1/3(738-750)) is necessary and sufficient to target a constitutively secreted protein to granules, and that L(745) anchors a hydrophobic patch that is critical for sorting. Also, we demonstrate that calcium binding by the second helix of PC1/3(711-753) promotes aggregation of the domain via the hydrophobic patch centered on L(745). These results provide a structure-function analysis of a DCSG-sorting domain, and reveal the importance of a hydrophobic patch and calcium binding in controlling the sorting of proteins containing alpha helices to DCSGs.
- Laboratory of Molecular Biochemistry of Hypertension, Clinical Research Institute of Montréal, Montréal, QC, Canada H2W 1R7.
Organizational Affiliation: