Senp1 Native Structure
Dong, C., Naismith, J.H.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| SENTRIN-SPECIFIC PROTEASE 1 | 226 | Homo sapiens | Mutation(s): 0 EC: 3.4.22 | 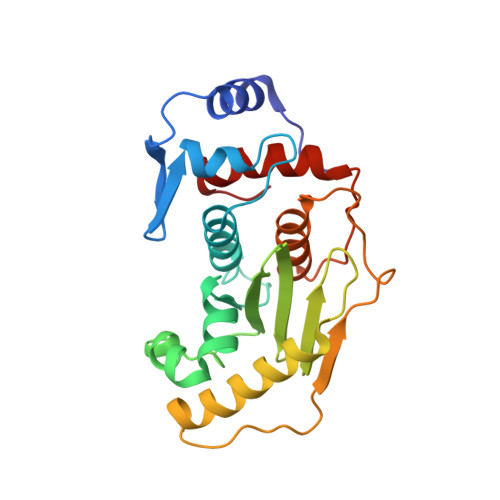 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9P0U3 GTEx: ENSG00000079387 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9P0U3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| SMALL UBIQUITIN-RELATED MODIFIER 2 | 81 | Homo sapiens | Mutation(s): 0 | 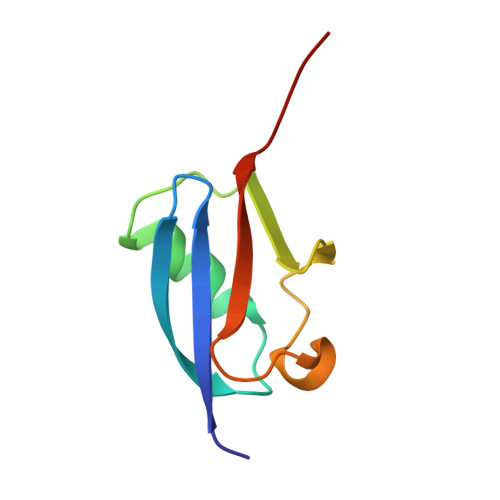 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61956 GTEx: ENSG00000188612 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61956 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 143.376 | α = 90 |
| b = 143.376 | β = 90 |
| c = 71.886 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |
| CCP4 | phasing |