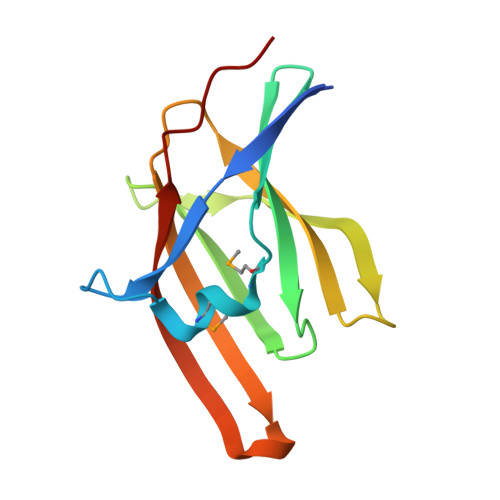
Haem recognition by a Staphylococcus aureus NEAT domain.
Grigg, J.C., Vermeiren, C.L., Heinrichs, D.E., Murphy, M.E.(2007) Mol Microbiol 63: 139-149
- PubMed: 17229211 Search on PubMed
- DOI: https://doi.org/10.1111/j.1365-2958.2006.05502.x
- Primary Citation Related Structures:
2ITE, 2ITF - PubMed Abstract:
Successful pathogenic organisms have developed mechanisms to thrive under extreme levels of iron restriction. Haem-iron represents the largest iron reservoir in the human body and is a significant source of iron for some bacterial pathogens. NEAT (NEAr Transporter) domains are found exclusively in a family of cell surface proteins in Gram-positive bacteria. Many NEAT domain-containing proteins, including IsdA in Staphylococcus aureus, are implicated in haem binding. Here, we show that overexpression of IsdA in S. aureus enhances growth and an inactivation mutant of IsdA has a growth defect, compared with wild type, when grown in media containing haem as the sole iron source. Furthermore, the haem-binding property of IsdA is contained within the NEAT domain. Crystal structures of the apo-IsdA NEAT domain and in complex with haem were solved and reveal a clathrin adapter-like beta-sandwich fold with a large hydrophobic haem-binding pocket. Haem is bound with the propionate groups directed at the molecular surface and the iron is co-ordinated solely by Tyr(166). The phenol groups of Tyr(166) and Tyr(170) form an H-bond that may function in regulating haem binding and release. An analysis of IsdA structure-sequence alignments indicate that conservation of Tyr(166) is a predictor of haem binding by NEAT domains.
- Department of Microbiology and Immunology, Life Sciences Institute, The University of British Columbia, Vancouver, BC, Canada V6T 1Z3.
Organizational Affiliation: