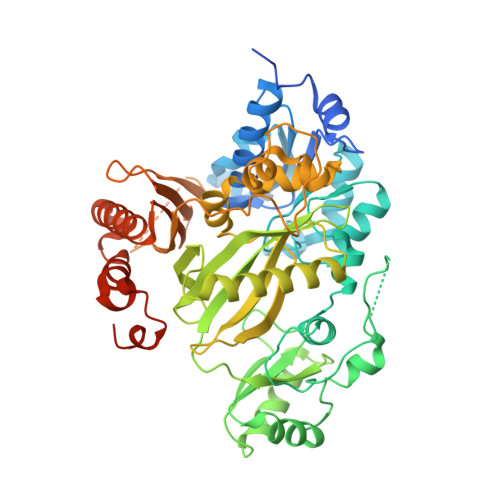
Crystal structure of the biotin carboxylase domain of human acetyl-CoA carboxylase 2.
Cho, Y.S., Lee, J.I., Shin, D., Kim, H.T., Cheon, Y.H., Seo, C.I., Kim, Y.E., Hyun, Y.L., Lee, Y.S., Sugiyama, K., Park, S.Y., Ro, S., Cho, J.M., Lee, T.G., Heo, Y.S.(2008) Proteins 70: 268-272
- PubMed: 17876819 Search on PubMed
- DOI: https://doi.org/10.1002/prot.21611
- Primary Citation Related Structures:
2HJW - R&D Center, CrystalGenomics, Inc., Seoul 138-739, Korea.
Organizational Affiliation: