Structural basis for the altered regulatory roles of three LysR-type transcritional variants of CatM and BenM: CatM(V158M), CatM(R156H) and BenM(R156H/T157S)
Ezezika, O.C., Neidle, E.L., Momany, C.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| HTH-type transcriptional regulator catM | 313 | Acinetobacter baylyi ADP1 | Mutation(s): 1 Gene Names: catM, catR | 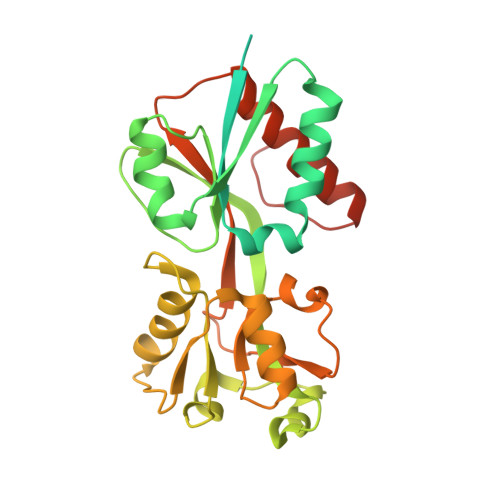 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P07774 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CL Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 37.447 | α = 69.55 |
| b = 51.038 | β = 89.51 |
| c = 59.12 | γ = 77.63 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| MOLREP | phasing |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| HKL-2000 | data scaling |