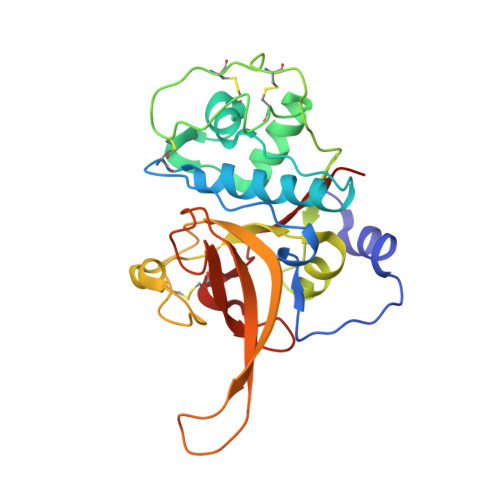
Structural and Functional Characterization of Falcipain-2, a Hemoglobinase from the Malarial Parasite Plasmodium falciparum.
Hogg, T., Nagarajan, K., Herzberg, S., Chen, L., Shen, X., Jiang, H., Wecke, M., Blohmke, C., Hilgenfeld, R., Schmidt, C.L.(2006) J Biological Chem 281: 25425-25437
- PubMed: 16777845 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M603776200
- Primary Citation Related Structures:
2GHU - PubMed Abstract:
Malaria is caused by protozoan erythrocytic parasites of the Plasmodium genus, with Plasmodium falciparum being the most dangerous and widespread disease-causing species. Falcipain-2 (FP-2) of P. falciparum is a papain-family (C1A) cysteine protease that plays an important role in the parasite life cycle by degrading erythrocyte proteins, most notably hemoglobin. Inhibition of FP-2 and its paralogues prevents parasite maturation, suggesting these proteins may be valuable targets for the design of novel antimalarial drugs, but lack of structural knowledge has impeded progress toward the rational discovery of potent, selective, and efficacious inhibitors. As a first step toward this goal, we present here the crystal structure of mature FP-2 at 3.1 A resolution, revealing novel structural features of the FP-2 subfamily proteases including a dynamic beta-hairpin hemoglobin binding motif, a flexible N-terminal alpha-helical extension, and a unique active-site cleft. We also demonstrate by biochemical methods that mature FP-2 can proteolytically process its own precursor in trans at neutral to weakly alkaline pH, that the binding of hemoglobin to FP-2 is strictly pH-dependent, and that FP-2 preferentially binds methemoglobin over hemoglobin. Because the specificity and proteolytic activity of FP-2 toward its multiple targets appears to be pH-dependent, we suggest that environmental pH may play an important role in orchestrating FP-2 function over the different life stages of the parasite. Moreover, it appears that selectivity of FP-2 for methemoglobin may represent an evolutionary adaptation to oxidative stress conditions within the host cell.
- Institute of Biochemistry, Center for Structural and Cell Biology in Medicine, University of Lübeck, Ratzeburger Allee 160, 23538 Lübeck, Germany.
Organizational Affiliation: