Insight into the radical mechanism of phycocyanobilin-ferredoxin oxidoreductase (PcyA) revealed by X-ray crystallography and biochemical measurements.
Tu, S.L., Rockwell, N.C., Lagarias, J.C., Fisher, A.J.(2007) Biochemistry 46: 1484-1494
- PubMed: 17279614 Search on PubMed
- DOI: https://doi.org/10.1021/bi062038f
- Primary Citation Related Structures:
2G18 - PubMed Abstract:
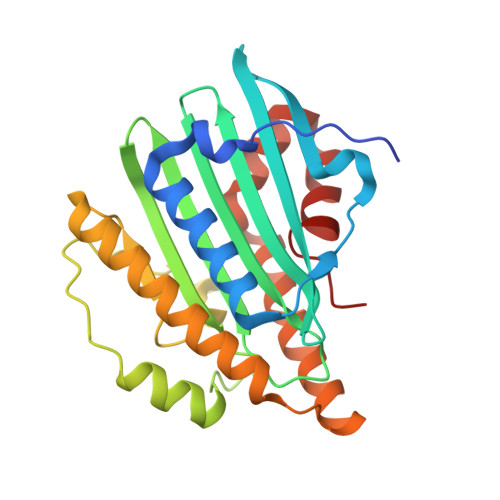
The X-ray crystal structure of the substrate-free form of phycocyanobilin (PCB)-ferredoxin oxidoreductase (PcyA; EC 1.3.7.5) from the cyanobacterium Nostoc sp. PCC7120 has been solved at 2.5 A resolution. A comparative analysis of this structure with those recently reported for substrate-bound and substrate-free forms of PcyA from the cyanobacterium Synechocystis sp. PCC6803 (Hagiwara et al. (2006) Proc. Natl. Acad. Sci. U.S.A. 103, 27-32; Hagiwara et al. (2006) FEBS Lett. 580, 3823-3828) provides a compelling picture of substrate-induced changes in the PcyA enzyme and the chemical basis of PcyA's catalytic activity. On the basis of these structures and the biochemical analysis of site-directed mutants of Nostoc PcyA, including mutants reported in recent studies (Tu et al. (2006) J. Biol. Chem. 281, 3127-3136) as well as mutants described in this study, a revised mechanism for the PcyA-mediated four-electron reduction of biliverdin IXalpha to 3E/3Z-phycocyanobilin via enzyme-bound bilin radical intermediates is proposed. The mechanistic insight of these studies, along with homology modeling, have provided new insight into the catalytic mechanisms of other members of the ferredoxin-dependent bilin reductase family that are widespread in oxygenic photosynthetic organisms.
- Institute of Plant and Microbial Biology, Academia Sinica, Nankang, Taipei 115, Taiwan.
Organizational Affiliation: