Human Cathepsin S with Inhibitor CRA-26871
Somoza, J.R.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| cathepsin S | 225 | Homo sapiens | Mutation(s): 0 EC: 3.4.22.27 | 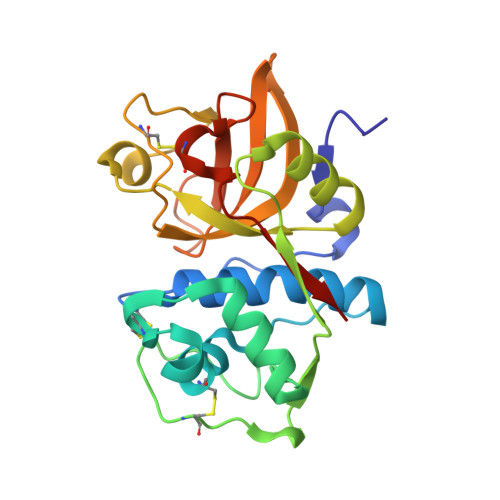 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P25774 GTEx: ENSG00000163131 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P25774 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| C71 Download:Ideal Coordinates CCD File | C [auth A], D [auth B] | N-[4-(AMINOMETHYL)-1,1-DIOXIDOTETRAHYDRO-2H-THIOPYRAN-4-YL]-3-(1-METHYLCYCLOPENTYL)-N~2~-[(1E)-N-(PHENYLSULFONYL)ETHANIMIDOYL]-L-ALANINAMIDE C23 H36 N4 O5 S2 HHATUZWPRPHWDY-FQEVSTJZSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 85.886 | α = 90 |
| b = 85.886 | β = 90 |
| c = 151.488 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| CNS | refinement |
| PDB_EXTRACT | data extraction |
| EPMR | phasing |