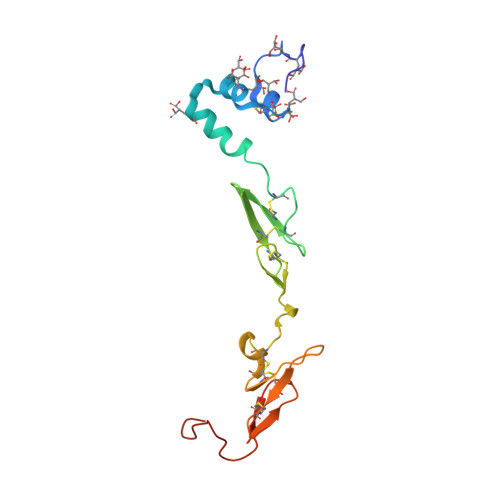
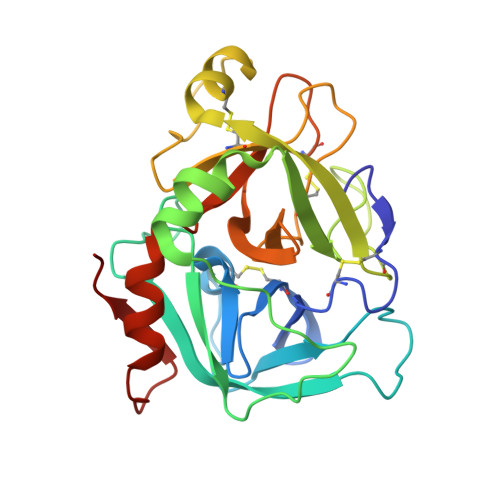
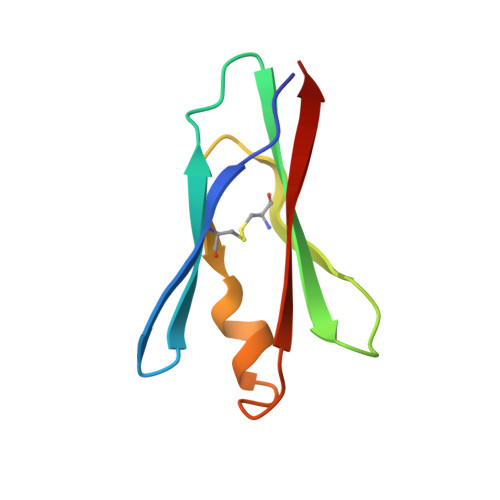
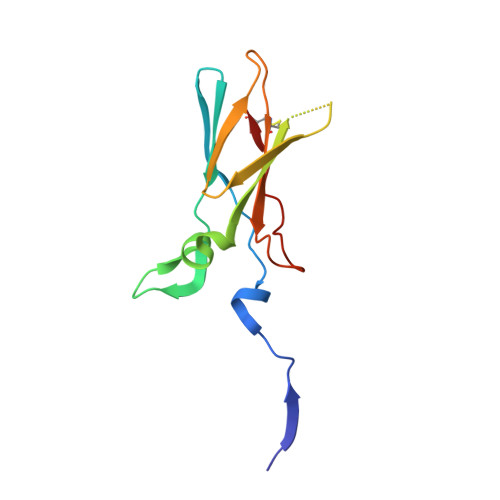
Probing the S2 site of factor VIIa to generate potent and selective inhibitors: the structure of BCX-3607 in complex with tissue factor-factor VIIa.
Krishnan, R., Kotian, P.L., Chand, P., Bantia, S., Rowland, S., Babu, Y.S.(2007) Acta Crystallogr D Biol Crystallogr 63: 689-697
- PubMed: 17505107 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444907014187
- Primary Citation Related Structures:
2EC9 - PubMed Abstract:
Factor VIIa (FVIIa) is a trypsin-like serine protease in the coagulation cascade. Its complex with tissue factor (TF) triggers the extrinsic pathway of the coagulation cascade, generating a blood clot. Research programs at several centers now recognize the important roles played by TF and FVIIa in both the thrombotic and inflammatory processes associated with cardiovascular diseases. Therefore, inhibition of the TF-FVIIa complex is seen as a promising target that is key to the development of clinical candidates for various cardiovascular applications. The crystal structure of the TF-FVIIa enzyme complex has been analyzed in order to design and synthesize small-molecule inhibitors. Using structure-based drug design (SBDD), a new series of inhibitors have been discovered that demonstrate high potency against the TF-FVIIa complex while maintaining substantial selectivity versus other closely related serine proteases such as trypsin, thrombin, factor Xa and plasmin.
- BioCryst Pharmaceuticals, 2190 Parkway Lake Drive, Birmingham, AL 35216, USA. rkrishnan@biocryst.com
Organizational Affiliation: