Active site plasticity of endonuclease VIII: Conformational changes compensating for different substrate and mutations of critical residues
Golan, G., Zharkov, D.O., Grollman, A.P., Shoham, G.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Endonuclease VIII | C [auth A] | 262 | Escherichia coli | Mutation(s): 0 Gene Names: nei EC: 3.2.2 (PDB Primary Data), 4.2.99.18 (PDB Primary Data) | 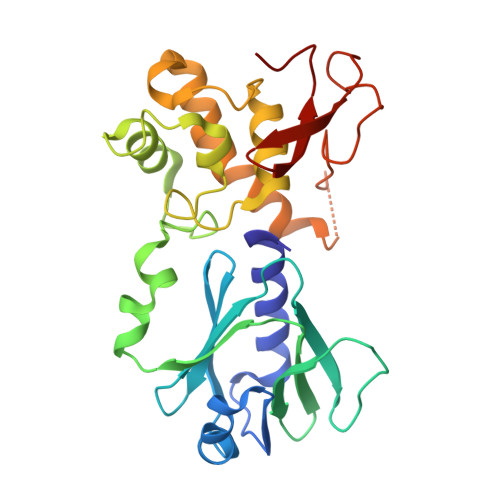 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P50465 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 1 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| 5'-D(P*GP*GP*CP*TP*TP*CP*AP*TP*CP*CP*TP*G)-3' | A [auth B] | 12 | N/A | 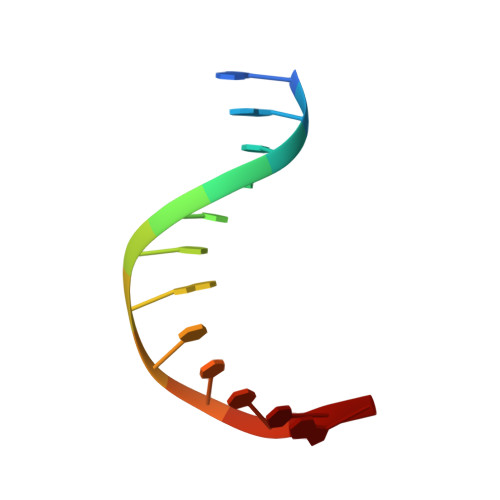 |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| 5'-D(P*CP*AP*GP*GP*AP*(PED)P*GP*AP*AP*GP*CP*C)-3' | B [auth C] | 12 | N/A | 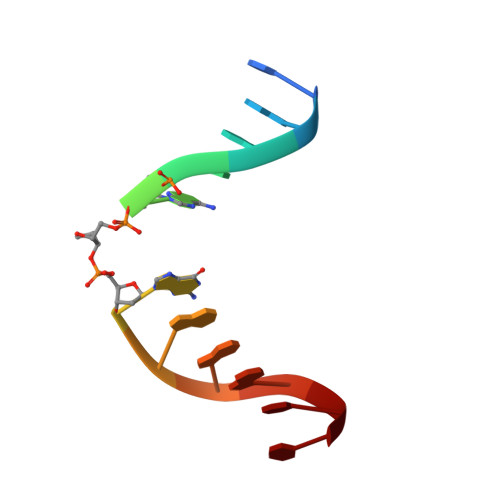 |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Download:Ideal Coordinates CCD File | D [auth C], F [auth A], G [auth A], H [auth A], I [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | J [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | E [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 76.184 | α = 90 |
| b = 76.184 | β = 90 |
| c = 164.54 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SHELX | model building |
| SHELXL-97 | refinement |
| ADSC | data collection |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| SHELX | phasing |