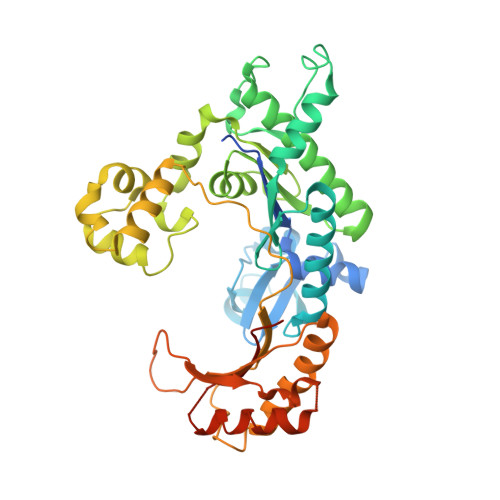
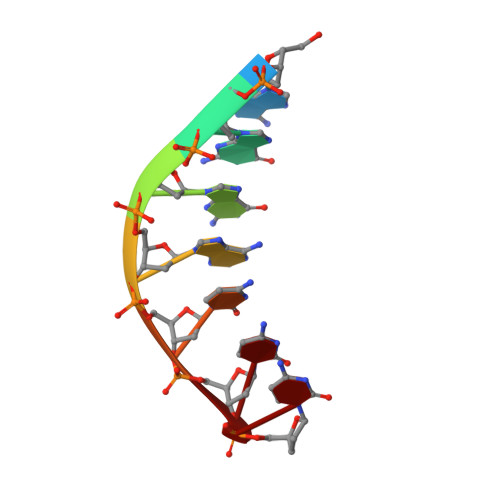
Hoogsteen base pair formation promotes synthesis opposite the 1,N(6)-ethenodeoxyadenosine lesion by human DNA polymerase iota.
Nair, D.T., Johnson, R.E., Prakash, L., Prakash, S., Aggarwal, A.K.(2006) Nat Struct Mol Biol 13: 619-625
- PubMed: 16819516 Search on PubMed
- DOI: https://doi.org/10.1038/nsmb1118
- Primary Citation Related Structures:
2DPI, 2DPJ - PubMed Abstract:
The 1,N6-ethenodeoxyadenosine (epsilon dA) lesion is promutagenic and has been implicated in carcinogenesis. We show here that human Pol iota, a Y-family DNA polymerase, can promote replication through this lesion by proficiently incorporating a nucleotide opposite it. The structural basis of this action is rotation of the epsilon dA adduct to the syn conformation in the Pol iota active site and presentation of its 'Hoogsteen edge' for hydrogen-bonding with incoming dTTP or dCTP. We also show that Pol zeta carries out the subsequent extension reaction and that efficiency of extension from epsilon dA x T is notably higher than from epsilon dA x C. Together, our studies reveal for the first time how the exocyclic epsilon dA adduct is accommodated in a DNA polymerase active site, and they show that the combined action of Pol iota and Pol zeta provides for efficient and error-free synthesis through this potentially carcinogenic DNA lesion.
- Department of Molecular Physiology and Biophysics, Mount Sinai School of Medicine, Box 1677, 1425 Madison Avenue, New York, New York 10029, USA.
Organizational Affiliation: