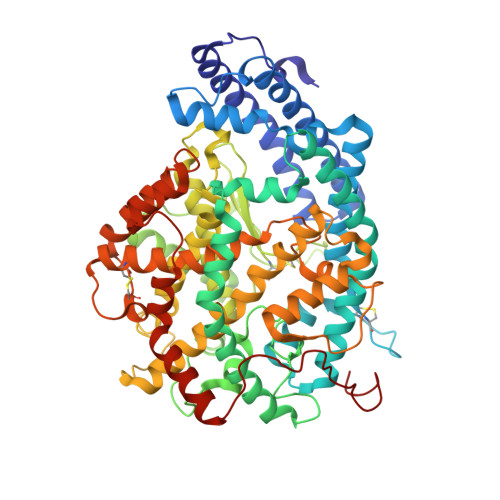
Crystal Structure of the N Domain of Human Somatic Angiotensin I-Converting Enzyme Provides a Structural Basis for Domain-Specific Inhibitor Design.
Corradi, H.R., Schwager, S.L.U., Nchinda, A.T., Sturrock, E.D., Acharya, K.R.(2006) J Mol Biology 357: 964
- PubMed: 16476442 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2006.01.048
- Primary Citation Related Structures:
2C6F, 2C6N - PubMed Abstract:
Human somatic angiotensin I-converting enzyme (sACE) is a key regulator of blood pressure and an important drug target for combating cardiovascular and renal disease. sACE comprises two homologous metallopeptidase domains, N and C, joined by an inter-domain linker. Both domains are capable of cleaving the two hemoregulatory peptides angiotensin I and bradykinin, but differ in their affinities for a range of other substrates and inhibitors. Previously we determined the structure of testis ACE (C domain); here we present the crystal structure of the N domain of sACE (both in the presence and absence of the antihypertensive drug lisinopril) in order to aid the understanding of how these two domains differ in specificity and function. In addition, the structure of most of the inter-domain linker allows us to propose relative domain positions for sACE that may contribute to the domain cooperativity. The structure now provides a platform for the design of "domain-specific" second-generation ACE inhibitors.
- Department of Biology and Biochemistry, University of Bath, Claverton Down, Bath BA2 7AY, UK.
Organizational Affiliation: