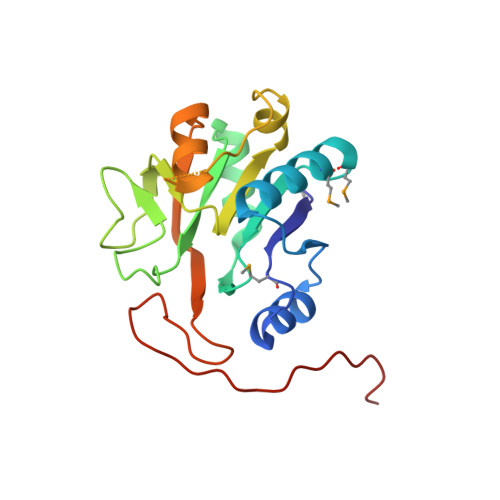
Structure of A197 from Sulfolobus Turreted Icosahedral Virus: A Crenarchaeal Viral Glycosyltransferase Exhibiting the Gt-A Fold.
Larson, E.T., Reiter, D., Young, M., Lawrence, C.M.(2006) J Virol 80: 7636
- PubMed: 16840342 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.00567-06
- Primary Citation Related Structures:
2C0N - PubMed Abstract:
Sulfolobus turreted icosahedral virus (STIV) was the first icosahedral virus characterized from an archaeal host. It infects Sulfolobus species that thrive in the acidic hot springs (pH 2.9 to 3.9 and 72 to 92 degrees C) of Yellowstone National Park. The overall capsid architecture and the structure of its major capsid protein are very similar to those of the bacteriophage PRD1 and eukaryotic viruses Paramecium bursaria Chlorella virus 1 and adenovirus, suggesting a viral lineage that predates the three domains of life. The 17,663-base-pair, circular, double-stranded DNA genome contains 36 potential open reading frames, whose sequences generally show little similarity to other genes in the sequence databases. However, functional and evolutionary information may be suggested by a protein's three-dimensional structure. To this end, we have undertaken structural studies of the STIV proteome. Here we report our work on A197, the product of an STIV open reading frame. The structure of A197 reveals a GT-A fold that is common to many members of the glycosyltransferase superfamily. A197 possesses a canonical DXD motif and a putative catalytic base that are hallmarks of this family of enzymes, strongly suggesting a glycosyltransferase activity for A197. Potential roles for the putative glycosyltransferase activity of A197 and their evolutionary implications are discussed.
- Department of Chemistry and Biochemistry, Montana State University, Bozeman, MT 59715, USA.
Organizational Affiliation: