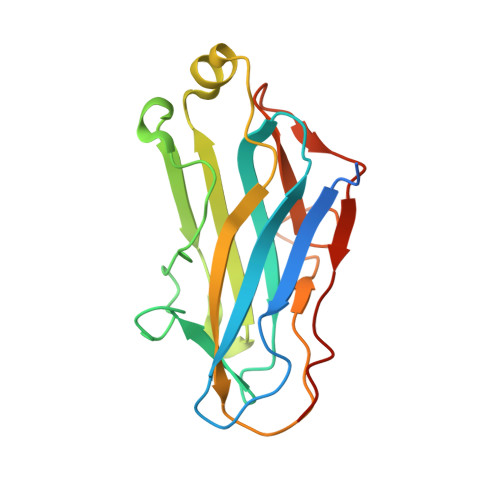
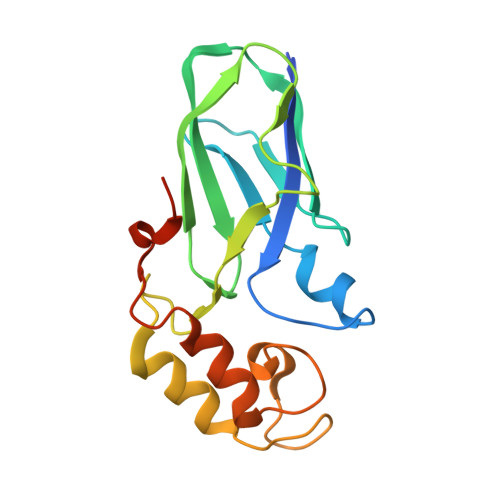
Mechanism of bacterial cell-surface attachment revealed by the structure of cellulosomal type II cohesin-dockerin complex.
Adams, J.J., Pal, G., Jia, Z., Smith, S.P.(2006) Proc Natl Acad Sci U S A 103: 305-310
- PubMed: 16384918 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0507109103
- Primary Citation Related Structures:
2B59 - PubMed Abstract:
Bacterial cell-surface attachment of macromolecular complexes maintains the microorganism in close proximity to extracellular substrates and allows for optimal uptake of hydrolytic byproducts. The cellulosome is a large multienzyme complex used by many anaerobic bacteria for the efficient degradation of plant cell-wall polysaccharides. The mechanism of cellulosome retention to the bacterial cell surface involves a calcium-mediated protein-protein interaction between the dockerin (Doc) module from the cellulosomal scaffold and a cohesin (Coh) module of cell-surface proteins located within the proteoglycan layer. Here, we report the structure of an ultra-high-affinity (K(a) = 1.44 x 10(10) M(-1)) complex between type II Doc, together with its neighboring X module from the cellulosome scaffold of Clostridium thermocellum, and a type II Coh module associated with the bacterial cell surface. Identification of X module-Doc and X module-Coh contacts reveal roles for the X module in Doc stability and enhanced Coh recognition. This extremely tight interaction involves one face of the Coh and both helices of the Doc and comprises significant hydrophobic character and a complementary extensive hydrogen-bond network. This structure represents a unique mechanism for cell-surface attachment in anaerobic bacteria and provides a rationale for discriminating between type I and type II Coh modules.
- Department of Biochemistry and Protein Function Discovery Group, Queen's University, Kingston, ON, Canada K7L 3N6.
Organizational Affiliation: