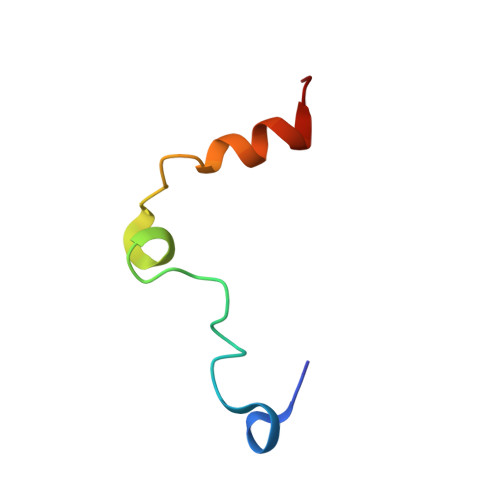
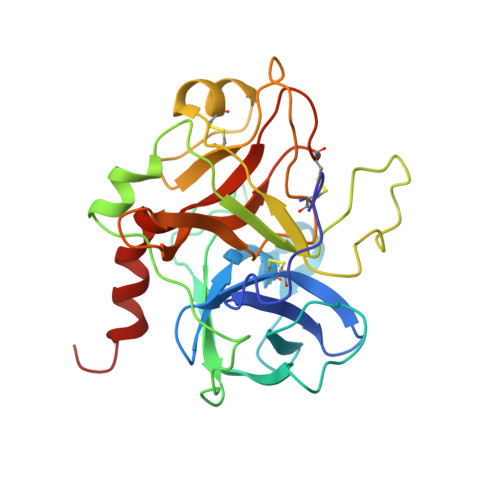
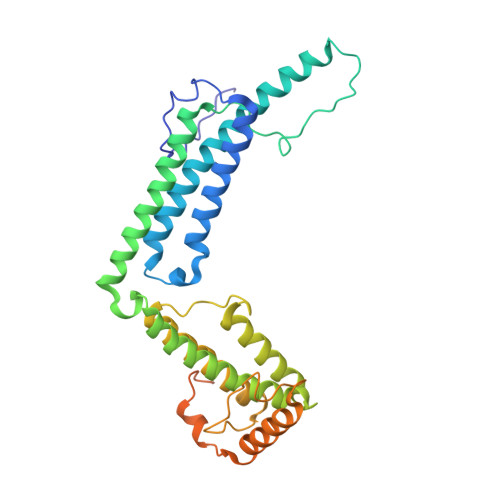
Structural Basis for Reduced Staphylocoagulase-mediated Bovine Prothrombin Activation
Friedrich, R., Panizzi, P., Kawabata, S., Bode, W., Bock, P.E., Fuentes-Prior, P.(2006) J Biological Chem 281: 1188-1195
- PubMed: 16230338 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M507957200
- Primary Citation Related Structures:
2A1D - PubMed Abstract:
Staphylocoagulase (SC) is a protein secreted by the human pathogen, Staphylococcus aureus, that activates human prothrombin (ProT) by inducing a conformational change. SC-bound ProT efficiently clots fibrinogen, thus bypassing the physiological blood coagulation pathway. The crystal structure of a fully active SC fragment, SC-(1-325), bound to human prethrombin 2 showed that the SC-(1-325) N terminus inserts into the Ile(16) pocket of prethrombin 2, thereby inducing expression of a functional catalytic site in the cognate zymogen without peptide bond cleavage. As shown here, SC-(1-325) binds to bovine and human ProT with similar affinity but activates the bovine zymogen only very poorly. By contrast to the approximately 2-fold difference in chromogenic substrate kinetic constants between human thrombin and the SC-(1-325).human (pro)thrombin complexes, SC-(1-325).bovine ProT shows a 3,500-fold lower k(cat)/K(m) compared with free bovine thrombin, because of a 47-fold increase in K(m) and a 67-fold decrease in k(cat). The SC-(1-325).bovine ProT complex is approximately 5,800-fold less active compared with its human counterpart. Comparison of human and bovine fibrinogen as substrates of human and bovine thrombin and the SC-(1-325).(pro)thrombin complexes indicates that the species specificity of SC-(1-325) cofactor activity is determined primarily by differences in conformational activation of bound ProT. These results suggest that the catalytic site in the SC-(1-325).bovine ProT complex is incompletely formed. The current crystal structure of SC-(1-325).bovine thrombin reveals that SC would dock similarly to the bovine proenzyme, whereas the bovine (pro)thrombin-characteristic residues Arg(144) and Arg(145) would likely interfere with insertion of the SC N terminus, thus explaining the greatly reduced activation of bovine ProT.
- Proteinase Research Group, Max Planck Institute of Biochemistry, Martinsried, Germany.
Organizational Affiliation: