Novel Mechanisms of pH Sensitivity in Tuna Hemoglobin: A STRUCTURAL EXPLANATION OF THE ROOT EFFECT
Yokoyama, T., Chong, K.T., Miyazaki, G., Morimoto, H., Shih, D.T., Unzai, S., Tame, J.R., Park, S.Y.(2004) J Biological Chem 279: 28632-28640
- PubMed: 15117955
- DOI: https://doi.org/10.1074/jbc.M401740200
- Primary Citation Related Structures:
1V4U, 1V4W, 1V4X - PubMed Abstract:
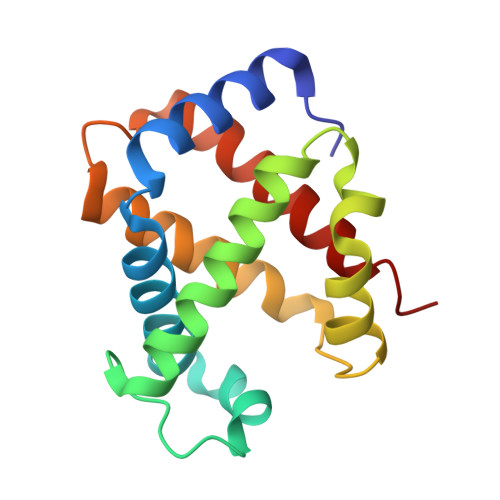
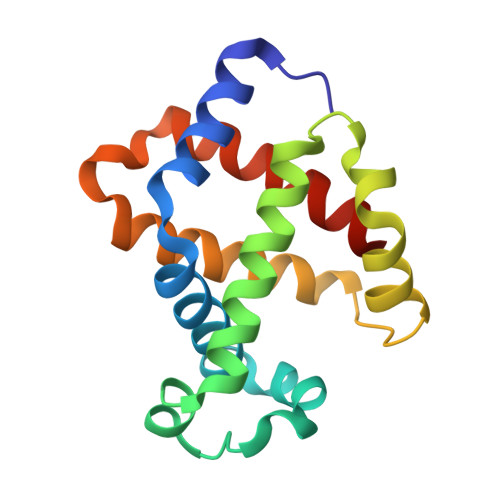
The crystal structure of hemoglobin has been known for several decades, yet various features of the molecule remain unexplained or controversial. Several animal hemoglobins have properties that cannot be readily explained in terms of their amino acid sequence and known atomic models of hemoglobin. Among these, fish hemoglobins are well known for their widely varying interactions with heterotropic effector molecules and pH sensitivity. Some fish hemoglobins are almost completely insensitive to pH (within physiological limits), whereas others show extremely low oxygen affinity under acid conditions, a phenomenon called the Root effect. X-ray crystal structures of Root effect hemoglobins have not, to date, provided convincing explanations of this effect. Sequence alignments have signally failed to pinpoint the residues involved, and site-directed mutagenesis has not yielded a human hemoglobin variant with this property. We have solved the crystal structure of tuna hemoglobin in the deoxy form at low and moderate pH and in the presence of carbon monoxide at high pH. A comparison of these models shows clear evidence for novel mechanisms of pH-dependent control of ligand affinity.
- Protein Design Laboratory, Yokohama City University, Suehiro-cho 1-7-29, Tsurumi, Yokohama 230-0045, Japan.
Organizational Affiliation: