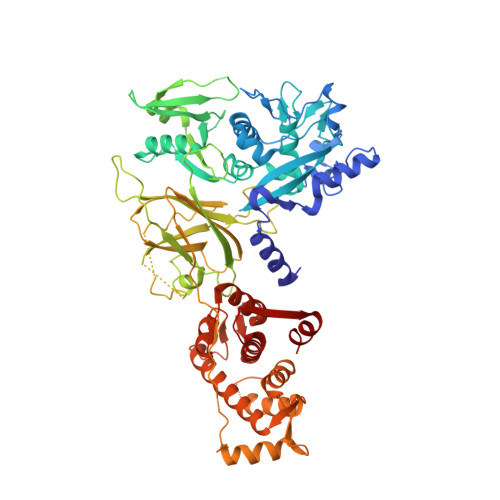
Activation of calpain by Ca2+: roles of the large subunit N-terminal and domain III-IV linker peptides
Hosfield, C.M., Elce, J.S., Jia, Z.(2004) J Mol Biology 343: 1049-1053
- PubMed: 15476820 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2004.08.073
- Primary Citation Related Structures:
1U5I - PubMed Abstract:
The calpains are a family of cysteine proteases with closely related amino acid sequences, but a wide range of Ca(2+) requirements (K(d)). For m-calpain, K(d) is approximately 325microM, for mu-calpain it is approximately 50microM, and for calpain 3 it is not strictly known but may be approximately 0.1microM. On the basis of previous structure determination of m-calpain we postulated that two regions of the calpain large subunits, the N-terminal peptide (residues 1-20) and a domain III-IV linker peptide (residues 514-530 in m-calpain) were important in defining K(d). The mutations Lys10Thr in the N-terminal peptide, and Glu517Pro in the domain linker peptide, reduced K(d) of m-calpain by 30% and 42%, respectively, revealing that these two regions are functionally important. The increased Ca(2+)-sensitivity of these mutants demonstrate that the Lys10-Asp148 salt link and the short beta-sheet interaction involving Glu517 are factors contributing to the high K(d) of m-calpain. Though these two regions are physically remote from the active site and Ca(2+)-binding site, they play significant roles in regulating the response of calpain to Ca(2+). Differences in these interactions in mu-calpain and in calpain 3 are also consistent with their progressively lower K(d) values.
- Department of Biochemistry, Queen's University, Kingston, Ont., Canada K7L 3N6.
Organizational Affiliation: