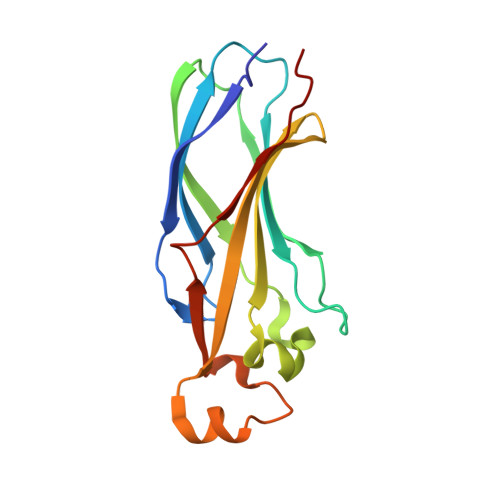
Structure and function of the conserved core of histone deposition protein Asf1.
Daganzo, S.M., Erzberger, J.P., Lam, W.M., Skordalakes, E., Zhang, R., Franco, A.A., Brill, S.J., Adams, P.D., Berger, J.M., Kaufman, P.D.(2003) Curr Biol 13: 2148-2158
- PubMed: 14680630 Search on PubMed
- DOI: https://doi.org/10.1016/j.cub.2003.11.027
- Primary Citation Related Structures:
1ROC - PubMed Abstract:
Asf1 is a ubiquitous eukaryotic histone binding and deposition protein that mediates nucleosome formation in vitro and is required for genome stability in vivo. Studies in a variety of organisms have defined Asf1's role as a histone chaperone during DNA replication through specific interactions with histones H3/H4 and the histone deposition factor CAF-I. In addition to its role in replication, conserved interactions with proteins involved in chromatin silencing, transcription, chromatin remodeling, and DNA repair have also established Asf1 as an important component of a number of chromatin assembly and modulation complexes. We demonstrate that the highly conserved N-terminal domain of S. cerevisiae Asf1 (Asf1N) is the core region that mediates all tested functions of the full-length protein. The crystal structure of this core domain, determined to 1.5 A resolution, reveals a compact immunoglobulin-like beta sandwich fold topped by three helical linkers. The surface of Asf1 displays a conserved hydrophobic groove flanked on one side by an area of strong electronegative surface potential. These regions represent potential binding sites for histones and other interacting proteins. The structural model also allowed us to interpret mutagenesis studies of the human Asf1a/HIRA interaction and to functionally define the region of Asf1 responsible for Hir1-dependent telomeric silencing in budding yeast. The evolutionarily conserved, N-terminal 155 amino acids of histone deposition protein Asf1 are functional in vitro and in vivo. This core region of Asf1 adopts a compact immunoglobulin-fold structure with distinct surface characteristics, including a Hir protein binding region required for gene silencing.
- Lawrence Berkeley National Laboratory, University of California-Berkeley, Berkeley, CA 94720, USA.
Organizational Affiliation: