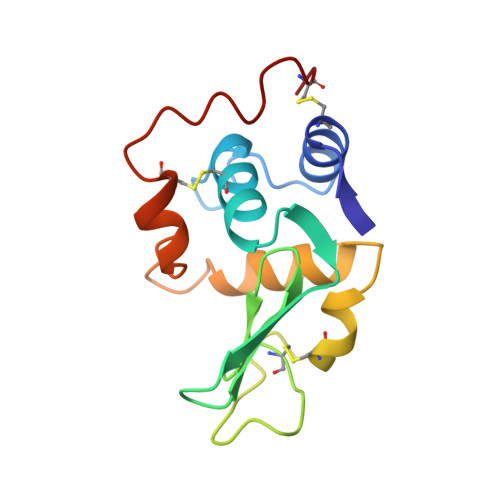
The crystal structure of a mutant human lysozyme C77/95A with increased secretion efficiency in yeast.
Inaka, K., Taniyama, Y., Kikuchi, M., Morikawa, K., Matsushima, M.(1991) J Biological Chem 266: 12599-12603
- PubMed: 2061330 Search on PubMed
- Primary Citation Related Structures:
1LHM - PubMed Abstract:
The three-dimensional structure of a mutant human lysozyme, C77/95A, in which residues Cys77 and Cys95 were replaced by alanine, was determined at 1.8-A resolution by x-ray crystallography. The properties of this mutant protein have been well characterized with respect to its thermal stability and secretion efficiency in a yeast expression system. The overall three-dimensional structure of C77/95A was found to be essentially identical to that of the wild-type human lysozyme, although the coordinates were shifted by more than 0.5 A and the thermal factors of the main-chain atoms were increased in the vicinity of residue 77. The reduction in thermal stability of this mutant has been previously explained by an increase in entropy of the unfolded state. In addition, a packing defect (cavity) produced by the removal of the disulfide bond was detected in the three-dimensional structure of C77/95A. This cavity can also be a reason why the stability of the protein is reduced because the free energy of the folded state could be expected to increase. The increased secretion efficiency cannot be due mainly to the three-dimensional structure, but may possibly be related to some event in the pathway of protein secretion. One of the possibilities might involve molecular flexibilities in the secondary or tertiary structure for lack of one of the disulfide bonds.
- Protein Engineering Research Institute, Osaka, Japan.
Organizational Affiliation: