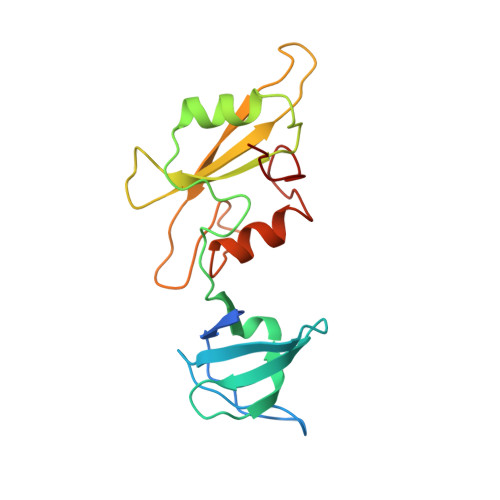
Structure of the regulatory domains of the Src-family tyrosine kinase Lck.
Eck, M.J., Atwell, S.K., Shoelson, S.E., Harrison, S.C.(1994) Nature 368: 764-769
- PubMed: 7512222 Search on PubMed
- DOI: https://doi.org/10.1038/368764a0
- Primary Citation Related Structures:
1LCK - PubMed Abstract:
The kinase p56lck (Lck) is a T-lymphocyte-specific member of the Src family of non-receptor tyrosine kinases. Members of the Src family each contain unique amino-terminal regions, followed by Src-homology domains SH3 and SH2, and a tyrosine kinase domain. SH3 and SH2 domains mediate critical protein interactions in many signal-transducing pathways. They are small, independently folded modules of about 60 and 100 residues, respectively, and they are often but not always found together in the same molecule. Like all nine Src-family kinases (reviewed in ref. 3), Lck is regulated by phosphorylation of a tyrosine in the short C-terminal tail of its catalytic domain. There is evidence that binding of the phosphorylated tail to the SH2 domain inhibits catalytic activity of the kinase domain and that the SH3 and SH2 domains may act together to effect this regulation. Here we report the crystal structures for a fragment of Lck bearing its SH3 and SH2 domains, alone and in complex with a phosphotyrosyl peptide containing the sequence of the Lck C-terminal regulatory tail. The latter complex represents the regulatory apparatus of Lck. The SH3-SH2 fragment forms similar dimers in both crystals, and the tail peptide binds at the intermolecular SH3/SH2 contact. The two structures show how an SH3 domain might recognize a specific target and suggest how dimerization could play a role in regulating Src-family kinases.
- Howard Hughes Medical Institute, Boston, Massachusetts.
Organizational Affiliation: