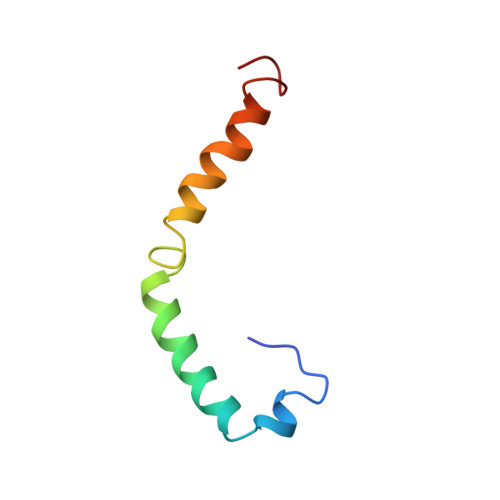
Conformation of human apolipoprotein C-I in a lipid-mimetic environment determined by CD and NMR spectroscopy.
Rozek, A., Sparrow, J.T., Weisgraber, K.H., Cushley, R.J.(1999) Biochemistry 38: 14475-14484
- PubMed: 10545169 Search on PubMed
- DOI: https://doi.org/10.1021/bi982966h
- Primary Citation Related Structures:
1IOJ - PubMed Abstract:
The high-resolution conformation of human apoC-I in complexes with sodium dodecyl sulfate (SDS) is presented. As estimated from CD data, apoC-I adopts 54% helical secondary structure when bound to SDS, which is similar to the helical content previously found with phospholipids. The NMR-derived conformation of apoC-I is composed of two amphipathic helices, residues 7-29 and 38-52, separated by a flexible linker. The N-terminal helix contains a mobile hinge involving residues 12-15. The hydrophobic side chains cluster on the nonpolar face of both helices, thus forming two discrete lipid-binding sites in the N-terminal helix and one in the C-terminal helix. As suggested by amide proton resonance line widths and deuterium exchange rates, the N-terminal helix is more flexible and may bind less tightly to the detergent than the C-terminal helix. The different mobility of both helices appears to be related to side-chain composition, rather than length of the amphipathic helix, and may play a role in the function of apoC-I as an activator of lecithin:cholesterol acyltransferase (LCAT). A model is suggested in which the C-terminal helix serves as a lipid anchor while the N-terminal helix may hinge off the lipid surface to make specific contacts with LCAT.
- Department of Chemistry and Institute of Molecular Biology and Biochemistry, Simon Fraser University, Burnaby, British Columbia Canada.
Organizational Affiliation: